Document Type : Original Research
Authors
- Fatemeh Khatami 1
- Bagher Larijani 2
- Ramin Heshmat 1
- Shirzad Nasiri 3
- Hiva Saffar 4
- Gita Shafiee 1
- Azam Mossafa 3
- Seyed Mohammad Tavangar 4
1 Chronic Diseases Research Center, Endocrinology and Metabolism Population Sciences Institute, Tehran University of Medical Sciences, Tehran, Iran
2 Endocrinology and Metabolism Research Center, Endocrinology and Metabolism Clinical Sciences Institute, Tehran University of Medical Sciences, Tehran, Iran
3 Department of Surgery, Tehran University of Medical Sciences, Shariati Hospital, Tehran, Iran
4 Department of Pathology, Shariati Hospital, Tehran University of Medical Sciences, Tehran, Iran
Abstract
Background & Objective:
Papillary thyroid cancer (PTC) is considered to be the most common type of thyroid malignancies. Epigenetic alteration, in which the chromatin conformation and gene expression change without changing the sequence of DNA, can occur in some tumor suppressor genes and oncogenes. Methylation is the most common type of epigenetic alterations that can be an excellent indicator of PTC invasive behavior.
Methods:
In this research, we determined the promoter methylation status of four tumor suppressor genes (SLC5A8, RASSF1, MGMT, and DNMT1) and compared the results of 55 PTC cases with 40 goiter patients. For methylation, we used the methylation-sensitive high resolution melting (MS-HRM) assay technique. The resulting graphs of each run were compared with those of 0%, 50%, and 100% methylated controls.
Result:
Our data showed that the promoter methylation of SLC5A8, Ras association domain family member 1(RASSF1), and MGMT were significantly different between PTC tissue and goiter with P-value less than 0.05. The most significant differences were observed in RASSF1; 77.2% of hyper-methylated PTC patients versus 15.6% hyper-methylated goiter samples (P<0.001).
Conclusion:
RASSF1 promoter methylation can be a PTC genetic marker. RASSF1 promoter methylation is under the impact of the methyltransferase genes (DNMT1 and MGMT), protein expression, and promoter methylation.
Highlights
✅RASSF1 promoter methylation can be a PTC genetic marker. RASSF1 promoter methylation is under the impact of the methyltransferase genes (DNMT1 and MGMT), protein expression, and promoter methylation.
Keywords
Main Subjects
Endocrine tumors include thyroid, adrenal, pancreas, parathyroid, and pituitary glands (1). Thyroid cancers are classified into four main types: papillary thyroid cancer (PTC), follicular thyroid cancer (FTC), medullary thyroid cancer (MTC), and anaplastic thyroid cancer (ATC) (2, 3). PTC is the most common form of well-differentiated thyroid cancer (1.0%–1.5% new cases per year) with growing incidence over the last three decades all over the world (4-8). PTC is typically an asymptomatic disease and it is identified through the mass in the anterior neck of patients usually when they are in their thirties and forties (9-11). PTC cells can occasionally migrate to the adjacent lymph nodes and rarely to distant organs; thus, adjacent and distant metastasis to the lungs and bones can be seen in metastatic form of PTC (12, 13). Nowadays, fine needle aspiration (FNA) is a common test for evaluating thyroid nodules, but it can be reported as the uncertain in some rare cases (14, 15). Accordingly, finding some genetic biomarkers for classifying malignant and benign cases before metastasis could be an essential measurement for both thyroid cancer patients and clinicians (16-19). In thyroid malignancies, the most important genetic and epigenetic alterations start their functions through activating metabolic pathways like mitogen-associated protein kinase (MAPK)/extracellular signal-regulated kinase (ERK) (20-22). Genetic and epigenetic changes of the genome can result in protein expression alterations like EGFRvIII, CD56, P63, CK19, estrogen receptors (ERs), and Survivin that are usually checked by immune-histochemical (IHC) studies (23-26). Histopathology is the microscopic study of targeted surgically removed tissue and for accurate diagnosis of cancer and other diseases, histopathological examination of samples is required (27-31). IHC is a useful method for determining the exact origin of tumor cells and sometimes discrimination between non-neoplastic disorders (32-35). Common histopathology and clinical features can be used for PTC and other papillary cancer types; moreover, some additional genetic and epigenetic biomarkers can support them (36). In fact, genetic and epigenetic biomarkers can fill the gap of exact diagnosis through imaging (ultrasound technology) and cytology, as the usual detection methods (37-39). Some genetic markers are mutations, polymorphisms, amplifications, and translocations, and epigenetic markers addition to microRNAs (34, 40). Contrary to genetic modifications that alter the sequence of genes constantly, the methylation is resulting in the alteration of gene expression patterns without changing the DNA sequence in a reversible manner (41, 42). Epigenetic silencing through aberrant DNA methylation of tumor suppressor genes can bring devastating consequences and cause human cancer formation (42, 43). Hyper-methylation of several tumor suppressor genes (TSHR, ECAD, SLC5A8, DAPK, TIMP3, and RARB2) are linked to the aggressive features of PTC (27, 44-47). Methylation status is mostly reported as the Methylated (M) or Unmethylated (U) so the methylation quantity is not available. Unfortunately, the common use of non-quantitative methylation detection method cannot represent the exact methylation in promoter region of reported hyper-methylated loci. Moreover, non-quantitative methylation detection method is prone to the inclusion of false positive results (48). Furthermore, the failure to quantify methylation incorrectly assumes homogeneity of stages and the significance of all detected methylation (49). Thanks to the new approach of promoter methylation quantification based on high resolution melting (HRM), now it is possible to determine the quantity of methylated cytosine in CpG dinucleotide (CpG islands) (50-53). Assessment of DNA methylation quantity can be a critical factor for the identification, development, and application of methylation-based biomarkers in cancer. This study aims to identify DNA methylation quantity of four tumor suppressor genes using the methylation-sensitive high resolution melting (MS-HRM) assay technique.
Materials and Methods
Tissue Samples
This study was approved by the Research Ethics Committee of the Endocrinology and Metabolism Research Institute, Tehran University of Medical Sciences (IR.TUMS.EMRI.REC.1395.00114). Totally, 95 human thyroid tissues were obtained from fresh frozen surgically resected thyroid tissues (≥15 mm). In order to reduce contamination, all resected tissues were snap frozen in liquid nitrogen and independently analyzed. The demographic information of the patients and adenoma characteristics analyzed in this study are presented in Table 1. An informed consent was taken from all participants for the tissue collection in compliance with our institutional guidelines.
DNA extraction
Fresh frozen tissue specimens were cut on dry ice from fresh frozen surgical material stored at −180°C by using a scalpel. Then, DNA was extracted using the DNeasy Blood & Tissue Kit (Qiagen, Cat No:69504) according to the manufacturer’s protocol. DNA purity and quantity was determined using a Thermo Scientific™ NanoDrop™ spectrophotometers 2000c spectrophotometer (Thermo Fisher Scientific Inc). All the extracted thyroid tissue DNAs were stored at - 80°C.
Table 1. Demographics of PTC patients (cases) and goiter patients (control)
| Variables | Case (n= 55) | Controls (n=40) | P-value |
| Age (years) | 42.28 (±14.32) | 43.16 (±9..31) | 0.624 |
| Gender | |||
| Female | 38 (70.90%) | 31 (77.50%) | |
| Male | 17 (30.90%) | 9 (22.5%) | 0.306 |
| Weight | 70.51 (±12.19) | 73.60 (±10.91) | 0.204 |
| Height | 164.96 (±7.96) | 167.32 (±9.55) | 0.370 |
| BMI | 28.85 (±4.09) | 31.26 (±0.79) | 0.093 |
| SBP | 121.91 (±16.01) | 118.77 (±11.10) | 0.963 |
| DBP | 76.58 (±11.79) | 70.05 (±9.79) | 0.491 |
BMI: Body Mass Index, SBP: Systolic Blood Pressure, DBP: Diastolic Blood Pressure.
There was no statistically significant difference between the two groups of PTC patients (cases) and goiter patients (control) in basic characteristics.
Bisulfite Modification and Quantitative Methylation Detection
Bisulfite conversion altered the DNA sequence depending on the methylation status of individual unmethylated Cytosines (C) conversion to uracil in genomic DNA; these changes can be detected via HRM analysis (54). Then, 150-200 ng of DNA from each sample was treated with sodium bisulfite conversion kit by the “EpiTect® Bisulfite Kit” (Qiagen, Cat No: 59104) according to the manufacturer's protocol. The melting profiles of bisulfite-modified PCR products can be used to indicate methylation status, when samples are unknown. These processes were run by bisulfiting pre-treatment and unbiased PCR amplification of both methylated and unmethylated templates of the targeted region. Therefore, for the MS-HRM of SLC5A8, RASSF1, MGMT and DNMT1 genes promoter region amplification we used gene specific primers. The MS-HRM analyses were run based on three main stages: holding stage, cycling stage, and melt curve stage.
Statistical Analysis
Samples were considered as hyper-methylated and hypo-methylated when the measured methylation point was more than the 12% mean methylation level and less than the 12% mean methylation level, respectively. Correlations between the methylation levels and demographic and histopathological characteristics in the two groups were analyzed using DNA methylation. All analyses were done by SPSS®, version 16.0, license (SPSS Inc., Chicago, IL. USA), and P-value
Results
We examined the 12-loci promoter methylation of fresh frozen tissue (55 PTC cases vs. 40 goiter controls) for four tumor suppressor genes. The age of all samples ranged from 18 to 86 years. For each run of MS-HRM, five wells were allocated to control samples (0%, 50%, and 100%). Numerous replicates of the diluted samples were amplified by PCR. Then melting profile of each reaction was used to define its methylation status. The hyper/hypo-methylations in each sample could be freely estimated by comparing them to the control peaks (Figure 1).

Fig. 1. The graph of targeted promoter region of RASSF1 in comparison with 50% controls. Part A indicates hypo-methylation (yellow line) and Part B indicates hyper-methylation (red line).
Quantification of each locus of the four targeted genes is presented in Table 3. In addition, each locus methylation quantification and the overall methylation status of each gene was earned through two regions of RASSF1, three regions of SLC5A8, three regions of DNMT1, and four regions of MGMT (Table 2).
The cut-off value of methylation was defined according to several references (U if it was ≤12% and M if it was ≥12%) (55-57).
All candidate tumor suppressor gene promoter hyper-methylation was significantly different in two groups of PTC cases and controls except DNMT1. Thus, for sensitivity analysis adjusting for demographic variables and risk factors we have done several logistic regression models for potentially confounding variables Table 3). Two DNA methyltransferases of MGMT and DNMT1 also adjusted in model III and IV in order to check their impact on the methylation pattern.
In model III (MGMT adjusted model) the odds ratio of RASSF1 and SLC5A8 promoter hyper-methylation and risk of PTC increased in comparison with model I (crude model). However, in model IV (DNMT1 adjusted model), the RASSF1 and SLC5A8 promoter hyper-methylation and risk of PTC decreased a little in comparison with model I (crude model).
Table 2. The difference of methylation quantification of 12-promoter loci of four targeted tumor suppressor genes
| Promoter Region | Methylation* | PTC Cases Number (percent) |
Goiter Cases Number (percent) |
P-value |
| SLC5A8 |
U | 16(29.09%) | 34(85.0%) | 0.002 |
| M | 39 (70.9%) | 8 (20.0%) | ||
| RASSF1 |
U | 11(20.8%) | 38 (95.0%) | <0.001** |
| M | 44(80.0%) | 2 (5.0%) | ||
| MGMT |
U | 7 (14.0%) | 29 (72.5%) | 0.001 |
| M | 49 (12.72%) | 11 (27.5%) | ||
| DNMT1 | U | 25 (45.45%) | 30 (75%) | 0.018 |
| M | 30 (54.54%) | 10 (25%) |
*Methylation is categorized from 1 to 5 according to methylation quantification results.
1=0 percent methylated; 2=25 percent methylated; 3=50 percent methylated; 4=75 percent methylated; and 5=100 percent methylated.
The mean methylation of several loci of one gene was calculated and categorized as U if it was ≤12% and M if it was ≥12%** The most significant P-value was reported for RASSF1 gene.
Table 3. The association of methylation in four tumor suppressor genes SLC5A8, RASSF1, MGMT, and DNMT1
| Model | SLC5A8 | RASSF1 |
| Model I | 8.24 (3.34 - 20.32) | 18.37 (6.65 - 50.76) |
| Model II | 8.72 (3.36 - 22.67) | 16.29 (5.72 - 46.34) |
| Model III | 9.94 (2.87 - 34.45) | 19.02 (4.97 - 75.52) |
| Model IV | 8.85 (3.23 - 24.24) | 16.52 (5.62 - 48.6) |
Model I is crude model, Model II is age and sex adjusted model, Model III is age, sex, and MGMT methylation status adjusted, and Model IV is age, sex, DNMT1 methylation status adjusted.
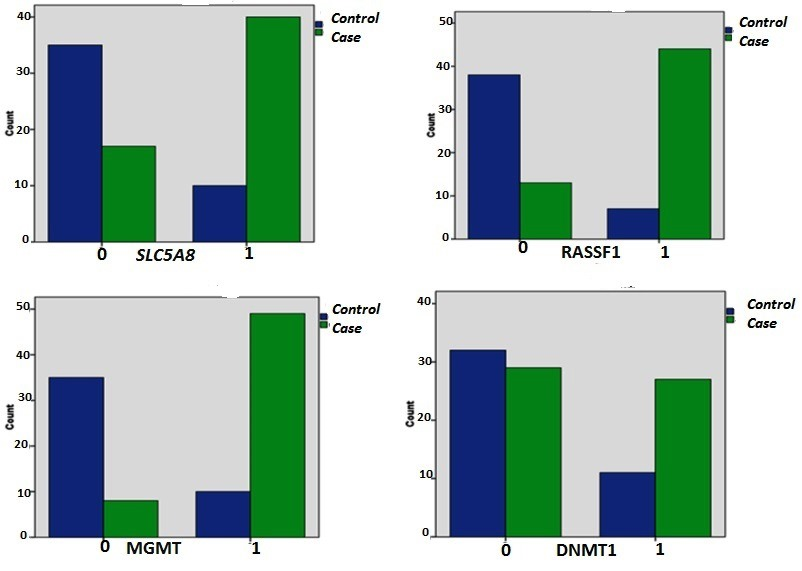
Fig. 2. Promoter quantification differences of SLC5A8, RASSF1, MGMT and DNMT1 methylation in PTC cases and controls.
Discussion
The role of aberrant methylation of tumor suppressor genes more than functionally chief regulatory genes is a frequent event in certain human tumors and developmental abnormalities (58, 59). In eukaryotic cells there are special DNA methyltransferase enzymes which put or remove the methyl group on/off the cytosine base of CpG islands and regulate the conformation change between methylated and unmethylated forms (60, 61). DNMT1 is the most abundant DNA methyltransferase in mammalian cells and extra active on hemi-methylated DNA as compared with unmethylated substrate in vitro, but it is still more active at de novo methylation than other DNMTs (62-64). Several studies have shown that changing the DNA methylation patterns in the regulatory promoter regions of DNMT1 play an important role in the development of genetic disorders (65-68). DNMT1 promoter methylation was present in primary and recurrent gliomas (69). The DNMT1 expression can be regulated through its promoter methylation patterns in the core promoter region of DNMT1 in several human neoplastic tissues (70, 71). The different methylation status of DNMT1 (b) in our study can be attributed to this region as the core promoter region that can have a role in PTC. It can also be supported by the idea that epigenetic regulation of the methylation status of DNMT genes can regulate epigenetic profile of extra embryonic tissue in humans (72). It has been shown that some medication targeting DNMT1 can inhibit migration and invasion of thyroid cancer cells through down-regulating DNMT1 (73). In contrast, some results indicated that DNMT1 was neither overexpressed in PTC nor correlated with tumor stage and capsular/vascular or lymphatic invasion (74). Yi Cai et al. (2017) pointed out to the critical threshold levels of DNMT1 as an important factor of DNA methylation maintenance across the genome in human cancer cells (75). However, our results indicated that DNMT1 methylation in PTC patients, in comparison with goiter patients (controls), was less than SLC5A8, RASSF1, and MGMT.
O6-methylguanine DNA methyltransferase is a protein in humans encoded by the MGMT gene, and it is a maintenance methyltransferases that is crucial for genome stability (76, 77). In a colorectal cancer study it was suggested that MGMT expression reduced after hyper-methylation of the MGMT promoter region (78). MGMT is suppressed epigenetically and in different ways such as promoter region hyper-methylation and over-expression of a number of microRNAs (79-84). Inactivation of the MGMT through promoter hyper-methylation is a common event in primary human malignancies (71, 85). Esteller et al. described a straight line of MGMT aberrant methylation and k-Ras and p53 genes mutation in colorectal cancer (85, 86). The promoter methylation of two candidate regions of MGMT were associated with PTC. In a MGMT adjusted model the odds ratio of RASSF1 and SLC5A8 promoter hyper-methylation and risk of PTC increased in comparison with model I (crude model). Supporting information is reported by Herfarth et al. that linked the specific CpG methylation pattern of the MGMT promoter region with decrease of MGMT expression in primary colorectal cancers (87). Moreover MGMT methylation is reported in a group of discriminating methylation markers that differentiate thyroid cancer from benign nodules (88). Moreover, MGMT methylation was reported in MLH1 and MGMT expression and their consequence in genomic instability in patients with thyroid carcinoma (89).
SLC5A8 can predominantly be found in the small intestine, colon, thyroid gland, kidney, and salivary glands and to a lesser extent in the retina and brain (90-93). It was shown that SLC5A8 expression, as a sodium/iodide symporter (NIS) member, decreased in several malignancies, including thyroid cancers and its methylation is shown as the discriminative marker between malignant and benign thyroid tumors (88, 94). We recently conducted a meta-analysis the results of which indicated that SLC5A8 was the most significant methylated gene in thyroid cancers (95). CpG island methylation of tumor-related promoters including RASSF1, MGMT, and SLC5A8 occurs preferentially in undifferentiated thyroid carcinoma (96). In contrary, the overexpression of SLC5A8 together with IRX1 and EBF3 may be involved in the transforming growth factor beta signaling pathway, which is often disrupted in head and neck squamous cell carcinoma. Silencing of the SLC5A8 through its promoter methylation was associated with BRAF mutations in classical PTC (97). Hyper-methylation of SLC5A8 promoters reported preferentially in undifferentiated carcinoma (98).
The Ras association domain-containing protein 1 (RASSF1), which is encoded by RAS gene altered expression, is associated with the pathogenesis of a variety of cancers (99-103). The most frequent molecular mechanism for RASSF1 suppression in different malignancies is the ishypermethylation of its CpG-island promoter region (104-108). Frequent epigenetic silencing of the RASSF1A in thyroid carcinoma has been highlithed (109) alone or with NORE1A methylation and BRAFV600E mutations (110). RASSF1 methylation can be used as the therapeutic determinant in thyroid malignancies (111). In a meta-analysis it was reported as the most significant hyper-methylated region within thyroid carcinomas (112). The survival rate and prognosis in head and neck squamous cell carcinoma (HNSCC) patients was dependent on 11 tumor-related genes, including RASSF1 and MGMT. The adjusted model of age, sex, and DNMT1 methylation status did not change the link of RASSF1 methylation and PTC cancer risk. Meanwhile, Bai et al. reported that DNMT1 inhibits proliferation, metastasis, and invasion in esophageal squamous cell carcinoma by suppressing methylation of RASSF1 and DAPK (30, 113). Methylation of RASSF1 gene promoter can be regulated by p53 and DAXX (114, 115).
According to the results, RASSF1 and SLC5A8 promoter methylations can be a PTC diagnostic biomarker which are completely dependent on DNMT1 or MGMT promoter methylation status.
Acknowledgements
The National Institute for Medical Research Development (NIMAD, Grant number: 965417) and Iran National Science Foundation (INSF, Grant number: 83107) funded this article.
Conflicts of Interest
The authors declared no conflict of interest regarding the publication of this article.
- Goodall C. On para‐endocrine cancer syndromes. International Journal of Cancer. 1969;4(1):1-13. [DOI:10.1002/ijc.2910040102]
- Sipos J, Mazzaferri E. Thyroid cancer epidemiology and prognostic variables. Clinical oncology. 2010;22(6):395-404. [DOI:10.1016/j.clon.2010.05.004] [PMID]
- Khatami F, Tavangar SM. Genetic and Epigenetic of Medullary Thyroid Cancer. Iranian biomedical journal. 2017:0-.
- Pellegriti G, Frasca F, Regalbuto C, Squatrito S, Vigneri R. Worldwide increasing incidence of thyroid cancer: update on epidemiology and risk factors. Journal of cancer epidemiology. 2013;2013. [DOI:10.1155/2013/965212] [PMID] [PMCID]
- Aschebrook-Kilfoy B, Ward MH, Sabra MM, Devesa SS. Thyroid cancer incidence patterns in the United States by histologic type, 1992-2006. Thyroid. 2011;21(2):125-34. [DOI:10.1089/thy.2010.0021] [PMID] [PMCID]
- Larijani B, Shirzad M, Mohagheghi M, Haghpanah V, Mosavi-Jarrahi A, Tavangar S, et al. Epidemiologic analysis of the Tehran cancer institute data system registry (TCIDSR). Asian Pac J Cancer Prev. 2004;5(1):36-9.
- Haghpanah V, Soliemanpour B, Heshmat R, Mosavi-Jarrahi A, Tavangar S, Malekzadeh R, et al. Endocrine cancer in Iran: based on cancer registry system. Indian journal of cancer. 2006;43(2):80. [DOI:10.4103/0019-509X.25889] [PMID]
- Larijani B, Mohagheghi MA, Bastanhagh MH, Mosavi-Jarrahi AR, Haghpanah V, Tavangar SM, et al. Primary thyroid malignancies in Tehran, Iran. Medical principles and practice. 2005;14(6):396-400. [DOI:10.1159/000088112] [PMID]
- Sosa JA, Udelsman R. Papillary thyroid cancer. Surgical Oncology Clinics. 2006;15(3):585-601. [DOI:10.1016/j.soc.2006.05.010] [PMID]
- Spartalis ED, Karatzas T, Charalampoudis P, Vergadis C, Dimitroulis D. Neglected papillary thyroid carcinoma seven years after initial diagnosis. Case reports in oncological medicine. 2013;2013. [DOI:10.1155/2013/148973] [PMID] [PMCID]
- Erden ES, Babayigit C, Davran R, Akin M, Karazincir S, Isaogullari N, et al. Papillary thyroid carcinoma with lung metastasis arising from dyshormonogenetic goiter: a case report. Case reports in medicine. 2013;2013. [DOI:10.1155/2013/813167] [PMID] [PMCID]
- Borschitz T, Eichhorn W, Fottner C, Hansen T, Schad A, Schadmand-Fischer S, et al. Diagnosis and treatment of pancreatic metastases of a papillary thyroid carcinoma. Thyroid. 2010;20(1):93-8. [DOI:10.1089/thy.2009.0026] [PMID] [PMCID]
- Sherman SI, Tielens ET, Sostre S, Wharam Jr M, Ladenson PW. Clinical utility of posttreatment radioiodine scans in the management of patients with thyroid carcinoma. The Journal of Clinical Endocrinology & Metabolism. 1994;78(3):629-34. https://doi.org/10.1210/jc.78.3.629 [DOI:10.1210/jcem.78.3.8126134]
- Haddadi-Nezhad S, Larijani B, Tavangar SM, Nouraei SM. Comparison of fine-needle-nonaspiration with fine-needle-aspiration technique in the cytologic studies of thyroid nodules. Endocrine pathology. 2003;14(4):369-73. [DOI:10.1385/EP:14:4:369]
- Greenblatt DY, Woltman T, Harter J, Starling J, Mack E, Chen H. Fine-needle aspiration optimizes surgical management in patients with thyroid cancer. Annals of surgical oncology. 2006;13(6):859-63. [DOI:10.1245/ASO.2006.08.020] [PMID]
- Grogan RH, Mitmaker EJ, Clark OH. The evolution of biomarkers in thyroid cancer-from mass screening to a personalized biosignature. Cancers. 2010;2(2):885-912. [DOI:10.3390/cancers2020885] [PMID] [PMCID]
- Sanii S, Saffar H, Tabriz HM, Qorbani M, Haghpanah V, Tavangar SM. Expression of matrix metalloproteinase-2, but not caspase-3, facilitates distinction between benign and malignant thyroid follicular neoplasms. Asian Pacific journal of cancer prevention. 2012;13(5):2175-8. [DOI:10.7314/APJCP.2012.13.5.2175] [PMID]
- Saffar H, Sanii S, Emami B, Heshmat R, Panah VH, Azimi S, et al. Evaluation of MMP2 and Caspase-3 expression in 107 cases of papillary thyroid carcinoma and its association with prognostic factors. Pathology-Research and Practice. 2013;209(3):195-9. [DOI:10.1016/j.prp.2012.06.011] [PMID]
- Haghpanah V, Lashkari A, Moradzadeh K, Tavangar SM. Hypereosinophilia as the presentation of metastatic medullary thyroid carcinoma: a remarkable event. The American journal of the medical sciences. 2007;334(2):131-2. [DOI:10.1097/MAJ.0b013e31812e 872f] [PMID]
- Xing M. BRAF mutation in thyroid cancer. Endocrine-related cancer. 2005;12(2):245-62. [DOI:10.1677/erc.1.0978] [PMID]
- Faam B, Ghaffari MA, Ghadiri ATA, Azizi F. Epigenetic modifications in human thyroid cancer. Biomedical Reports. 2015;3(1):3-8. [DOI:10.3892/br.2014.375] [PMID] [PMCID]
- Mohammadi-Asl J, Larijani B, Khorgami Z, Tavangar S, Haghpanah V, Mehdipour P. Prevalence of BRAFV600E mutation in Iranian patients with papillary thyroid carcinoma: a single-center study. J Appl Sci. 2009;9(19):3593-7. [DOI:10.3923/jas.2009.3593.3597]
- Omidfar K, Moinfar Z, Sohi AN, Tavangar SM, Haghpanah V, Heshmat R, et al. Expression of EGFRvIII in thyroid carcinoma: immunohistochemical study by camel antibodies. Immunological investigations. 2009;38(2):165-80. [DOI:10.1080/08820130902735998] [PMID]
- Haghpanah V, Shooshtarizadeh P, Heshmat R, Larijani B, Tavangar SM. Immunohistochemical analysis of survivin expression in thyroid follicular adenoma and carcinoma. Applied Immunohistochemistry & Molecular Morphology. 2006;14(4):422-5. [DOI:10.1097/01.pai.0000213100.880 74.b8] [PMID]
- El Demellawy D, Nasr A, Alowami S. Application of CD56, P63 and CK19 immunohistochemistry in the diagnosis of papillary carcinoma of the thyroid. Diagnostic pathology. 2008;3(1):5. [DOI:10.1186/1746-1596-3-5] [PMID] [PMCID]
- Tavangar S, Monajemzadeh M, Larijani B, Haghpanah V. Immunohistochemical study of oestrogen receptors in 351 human thyroid glands. Singapore medical journal. 2007;48(8):744-7.
- Amoli MM, Yazdani N, Amiri P, Sayahzadeh F, Haghpanah V, Tavangar SM, et al. HLA-DR association in papillary thyroid carcinoma. Disease markers. 2010;28(1):49-53. [DOI:10.1155/2010/130276] [PMID] [PMCID]
- Sabetkish S, Kajbafzadeh AM, Sabetkish N, Khorramirouz R, Akbarzadeh A, Seyedian SL, et al. Whole‐organ tissue engineering: decellularization and recellularization of three‐dimensional matrix liver scaffolds. Journal of biomedical materials research Part A. 2015;103(4):1498-508. [DOI:10.1002/jbm.a.35291] [PMID]
- Kajbafzadeh A-M, Payabvash S, Salmasi AH, Monajemzadeh M, Tavangar SM. Smooth muscle cell apoptosis and defective neural development in congenital ureteropelvic junction obstruction. The Journal of urology. 2006;176(2):718-23. [DOI:10.1016/j.juro.2006.03.041] [PMID]
- NASSERI‐MOGHADDAM S, Malekzadeh R, Sotoudeh M, Tavangar M, Azimi K, SOHRABPOUR AA, et al. Lower esophagus in dyspeptic Iranian patients: a prospective study. Journal of gastroenterology and hepatology. 2003;18(3):315-21. [DOI:10.1046/j.1440-1746.2003.02969.x] [PMID]
- Natanzi MM, Pasalar P, Kamalinejad M, Dehpour AR, Tavangar SM, Sharifi R, et al. Effect of aqueous extract of Elaeagnus angustifolia fruit on experimental cutaneous wound healing in rats. Acta Medica Iranica. 2012:589-96.
- Tavangar SM, Shojaee A, Tabriz HM, Haghpanah V, Larijani B, Heshmat R, et al. Immunohistochemical expression of Ki67, c-erbB-2, and c-kit antigens in benign and malignant pheochromocytoma. Pathology-Research and Practice. 2010;206(5):305-9. [DOI:10.1016/j.prp.2010.01.007] [PMID]
- Sarmadi S, Izadi-Mood N, Sotoudeh K, Tavangar SM. Altered PTEN expression; a diagnostic marker for differentiating normal, hyperplastic and neoplastic endometrium. Diagnostic pathology. 2009;4(1):41. [DOI:10.1186/1746-1596-4-41] [PMID] [PMCID]
- Alimoghaddam K, Shariftabrizi A, Tavangar M, Sanaat Z, Rostami S, Jahani M, et al. Anti-leukemic and anti-angiogenesis efficacy of arsenic trioxide in new cases of acute promyelocytic leukemia. Leukemia & lymphoma. 2006;47(1):81-8. [DOI:10.1080/10428190500300373] [PMID]
- Tavangar SM, Larijani B, Mahta A, Hosseini SMA, Mehrazine M, Bandarian F. Craniopharyngioma: a clinicopathological study of 141 cases. Endocrine pathology. 2004;15(4):339-44. [DOI:10.1385/EP:15:4:339]
- Khatami F, Mohammadamoli M, Tavangar SM. Genetic and epigenetic differences of benign and malignant pheochromocytomas and paragangliomas (PPGLs). Endocrine regulations. 2018;52(1):41-54. [DOI:10.2478/enr-2018-0006] [PMID]
- Hayes DF, Bast RC, Desch CE, Fritsche Jr H, Kemeny NE, Jessup JM, et al. Tumor marker utility grading system: a framework to evaluate clinical utility of tumor markers. JNCI: Journal of the National Cancer Institute. 1996;88(20):1456-66. [DOI:10.1093/jnci/88.20.1456] [PMID]
- Herceg Z, Hainaut P. Genetic and epigenetic alterations as biomarkers for cancer detection, diagnosis and prognosis. Molecular oncology. 2007;1(1):26-41. [DOI:10.1016/ j.molonc.2007.01.004] [PMID] [PMCID]
- Khatami F, Payab M, Sarvari M, Gilany K, Larijani B, Arjmand B, et al. Oncometabolites as biomarkers in thyroid cancer: a systematic review. Cancer Management and Research. 2019;11:1829. [DOI:10.2147/CMAR.S188661] [PMID] [PMCID]
- Kulasingam V, Diamandis EP. Strategies for discovering novel cancer biomarkers through utilization of emerging technologies. Nature Reviews Clinical Oncology. 2008;5(10):588. [DOI:10.1038/ncponc1187] [PMID]
- Xing M. Gene methylation in thyroid tumorigenesis. Endocrinology. 2007;148(3):948-53. [DOI:10.1210/en.2006-0927] [PMID]
- Phillips T. The role of methylation in gene expression. Nature Education. 2008;1(1):116.
- Suzuki MM, Bird A. DNA methylation landscapes: provocative insights from epigenomics. Nature Reviews Genetics. 2008;9(6):465. [DOI:10.1038/nrg2341] [PMID]
- Smith JA, Fan C-Y, Zou C, Bodenner D, Kokoska MS. Methylation status of genes in papillary thyroid carcinoma. Archives of Otolaryngology-Head & Neck Surgery. 2007;133(10):1006-11. [DOI:10.1001/archotol.133.10.1006] [PMID]
- Hu S, Liu D, Tufano RP, Carson KA, Rosenbaum E, Cohen Y, et al. Association of aberrant methylation of tumor suppressor genes with tumor aggressiveness and BRAF mutation in papillary thyroid cancer. International journal of cancer. 2006;119(10):2322-9. [DOI:10.1002/ijc.22110] [PMID]
- Xing M, Cohen Y, Mambo E, Tallini G, Udelsman R, Ladenson PW, et al. Early occurrence of RASSF1A hypermethylation and its mutual exclusion with BRAF mutation in thyroid tumorigenesis. Cancer research. 2004;64(5):1664-8. [DOI:10.1158/0008-5472.CAN-03-3242] [PMID]
- Mohammadi-asl J, Larijani B, Khorgami Z, Tavangar SM, Haghpanah V, Kheirollahi M, et al. Qualitative and quantitative promoter hypermethylation patterns of the P16, TSHR, RASSF1A and RARβ2 genes in papillary thyroid carcinoma. Medical oncology. 2011;28(4):1123-8. [DOI:10.1007/s12032-010-9587-z] [PMID]
- Lim AM, Candiloro IL, Wong N, Collins M, Do H, Takano EA, et al. Quantitative methodology is critical for assessing DNA methylation and impacts on correlation with patient outcome. Clinical epigenetics. 2014;6(1):22. [DOI:10.1186/1868-7083-6-22] [PMID] [PMCID]
- Mikeska T, Candiloro IL, Dobrovic A. The implications of heterogeneous DNA methylation for the accurate quantification of methylation. Epigenomics. 2010;2(4):561-73. [DOI:10.2217/epi.10.32] [PMID]
- Wittwer CT, Reed GH, Gundry CN, Vandersteen JG, Pryor RJ. High-resolution genotyping by amplicon melting analysis using LCGreen. Clinical chemistry. 2003;49(6):853-60. [DOI:10.1373/49.6.853] [PMID]
- Wojdacz TK, Dobrovic A. Methylation-sensitive high resolution melting (MS-HRM): a new approach for sensitive and high-throughput assessment of methylation. Nucleic acids research. 2007;35(6):e41. [DOI:10.1093/nar/gkm013] [PMID] [PMCID]
- Wojdacz TK, Dobrovic A, Hansen LL. Methylation-sensitive high-resolution melting. Nature protocols. 2008;3(12):1903. [DOI:10.1038/nprot.2008.191] [PMID]
- Hussmann D, Hansen LL. Methylation-Sensitive High Resolution Melting (MS-HRM). DNA Methylation Protocols: Springer; 2018. p. 551-71. [DOI:10.1007/978-1-4939-7481-8_28] [PMID]
- Dahl C, Grønskov K, Larsen LA, Guldberg P, Brøndum-Nielsen K. A homogeneous assay for analysis of FMR1 promoter methylation in patients with fragile X syndrome. Clinical chemistry. 2007;53(4):790-3. [DOI:10.1373/clinchem.2006.080762] [PMID]
- Brigliadori G, Foca F, Dall'Agata M, Rengucci C, Melegari E, Cerasoli S, et al. Defining the cutoff value of MGMT gene promoter methylation and its predictive capacity in glioblastoma. Journal of neuro-oncology. 2016;128(2):333-9. [DOI:10.1007/s11060-016-2116-y] [PMID]
- Quillien V, Lavenu A, Karayan‐Tapon L, Carpentier C, Labussière M, Lesimple T, et al. Comparative assessment of 5 methods (methylation‐specific polymerase chain reaction, methylight, pyrosequencing, methylation‐sensitive high‐resolution melting, and immunohistochemistry) to analyze O6‐methylguanine‐DNA‐methyltranferase in a series of 100 glioblastoma patients. Cancer. 2012;118(17):4201-11. [DOI:10.1002/ cncr.27392] [PMID]
- Xie H, Tubbs R, Yang B. Detection of MGMT promoter methylation in glioblastoma using pyrosequencing. International journal of clinical and experimental pathology. 2015;8(1):636.
- Esteller M, Corn PG, Baylin SB, Herman JG. A gene hypermethylation profile of human cancer. Cancer research. 2001;61(8):3225-9.
- Baylin SB. DNA methylation and gene silencing in cancer. Nature Reviews Clinical Oncology. 2005;2(S1):S4. [DOI:10.1038/ncponc0354] [PMID]
- Moore LD, Le T, Fan G. DNA Methylation and Its Basic Function. Neuropsychopharmacology. 2013;38(1):23-38. [DOI:10.1038/npp.2012.112] [PMID] [PMCID]
- Lopatina N, Haskell JF, Andrews LG, Poole JC, Saldanha S, Tollefsbol T. Differential maintenance and de novo methylating activity by three DNA methyltransferases in aging and immortalized fibroblasts. Journal of cellular biochemistry. 2002;84(2):324-34. [DOI:10.1002/jcb.10015] [PMID]
- Yokochi T, Robertson KD. Preferential methylation of unmethylated DNA by mammaliande Novo DNA methyltransferase Dnmt3a. Journal of Biological Chemistry. 2002;277(14):11735-45. [DOI:10.1074/jbc.M106590200] [PMID]
- Robert M-F, Morin S, Beaulieu N, Gauthier F, Chute IC, Barsalou A, et al. DNMT1 is required to maintain CpG methylation and aberrant gene silencing in human cancer cells. Nature genetics. 2003;33(1):61. [DOI:10.1038/ng1068] [PMID]
- Nuñez NN, Manlove AH, David SS. DNMT1 and Cancer: An Electrifying Link. Chemistry & biology. 2015;22(7):810-1. [DOI:10.1016/j.chembiol.2015.07.004] [PMID]
- Rahmani T, Azad M, Chahardouli B, Nasiri H, Vatanmakanian M, Kaviani S. Patterns of DNMT1 Promoter Methylation in Patients with Acute Lymphoblastic Leukemia. International journal of hematology-oncology and stem cell research. 2017; 11(3):172.
- Novakovic B, Wong NC, Sibson M, Ng H-K, Morley R, Manuelpillai U, et al. DNA methylation-mediated down-regulation of DNA methyltransferase-1 (DNMT1) is coincident with, but not essential for, global hypomethylation in human placenta. Journal of Biological Chemistry. 2010;285(13):9583-93. [DOI:10.1074/jbc.M109.064956] [PMID] [PMCID]
- Bennett KL, Karpenko M, Lin MT, Claus R, Arab K, Dyckhoff G, et al. Frequently methylated tumor suppressor genes in head and neck squamous cell carcinoma. Cancer Res. 2008;68(12):4494-9. [DOI:10.1158/0008-5472.CAN-07-6509] [PMID]
- Sharma S, Kelly TK, Jones PA. Epigenetics in cancer. Carcinogenesis. 2010;31(1):27-36. [DOI:10.1093/carcin/bgp220] [PMID] [PMCID]
- Gömöri É, Pál J, Kovács B, Dóczi T. Concurrent hypermethylation of DNMT1, MGMT and EGFR genes in progression of gliomas. Diagnostic pathology. 2012;7(1):8. [DOI:10.1186/1746-1596-7-8] [PMID] [PMCID]
- Li D, Bi F-F, Cao J-M, Cao C, Liu B, Yang Q. Regulation of DNA methyltransferase 1 transcription in BRCA1-mutated breast cancer: a novel crosstalk between E2F1 motif hypermethylation and loss of histone H3 lysine 9 acetylation. Molecular cancer. 2014;13(1):26. https://doi.org/10.1186/1476-4598-13-26 [DOI:10.1186/s12943-014-0285-x] [PMID] [PMCID]
- Khatami F, Mohebi SR, Ghiasi S, Haghighi MM, Safaee A, Hashemi M, et al. Amino acid substitution polymorphisms of two DNA methyltransferases and susceptibility to sporadic colorectal cancer. Gastroenterology and Hepatology from bed to bench. 2009;1(3).
- Cai Y, Tsai H-C, Yen R-WC, Zhang YW, Kong X, Wang W, et al. Critical threshold levels of DNA methyltransferase 1 are required to maintain DNA methylation across the genome in human cancer cells. Genome research. 2017;27(4):533-44. [DOI:10.1101/gr.208108.116] [PMID] [PMCID]
- Zhang Y, Sun B, Huang Z, Zhao D-W, Zeng Q. Shikonin Inhibites Migration and Invasion of Thyroid Cancer Cells by Downregulating DNMT1. Medical science monitor: international medical journal of experimental and clinical research. 2018;24:661. [DOI:10.12659/MSM.908381] [PMID] [PMCID]
- Brehar A, Procopiuc C, Paun D, Manda D, Oros S, Caragheorgheopol A, et al., editors. DNMT1 expression in papillary thyroid carcinoma. 16th European Congress of Endocrinology; 2014: BioScientifica. [DOI:10.1530/endoabs.35.P1099]
- Cai Y, Tsai H-C, Yen R-WC, Zhang YW, Kong X, Wang W, et al. Critical threshold levels of DNA methyltransferase 1 are required to maintain DNA methylation across the genome in human cancer cells. Genome research. 2017. [DOI:10.1101/gr.208108.116] [PMID] [PMCID]
- Tano K, Shiota S, Collier J, Foote RS, Mitra S. Isolation and structural characterization of a cDNA clone encoding the human DNA repair protein for O6-alkylguanine. Proceedings of the National Academy of Sciences. 1990;87(2):686-90. [DOI:10.1073/pnas.87.2.686] [PMID] [PMCID]
- Natarajan A, Vermeulen S, Darroudi F, Valentine MB, Brent TP, Mitra S, et al. Chromosomal localization of human O6-methylguanine-DNA methyltransferase (MGMT) gene by in situ hybridization. Mutagenesis. 1992;7(1):83-5. [DOI:10.1093/mutage/7.1.83] [PMID]
- Halford S, Rowan A, Sawyer E, Talbot I, Tomlinson I. O6-methylguanine methyltransferase in colorectal cancers: detection of mutations, loss of expression, and weak association with G: C> A: T transitions. Gut. 2005;54(6):797-802. [DOI:10.1136/gut.2004.059535] [PMID] [PMCID]
- CAbRINI G, FAbbRI E, Lo Nigro C, DEChECChI MC, GAMbARI R. Regulation of expression of O6-methylguanine-DNA methyltransferase and the treatment of glioblastoma. International journal of oncology. 2015;47(2):417-28. [DOI:10.3892/ijo.2015.3026] [PMID] [PMCID]
- Nakagawachi T, Soejima H, Urano T, Zhao W, Higashimoto K, Satoh Y, et al. Silencing effect of CpG island hypermethylation and histone modifications on O6-methylguanine-DNA methyltransferase (MGMT) gene expression in human cancer. Oncogene. 2003;22(55):8835. [DOI:10.1038/sj.onc.1207183] [PMID]
- Zhang W, Zhang J, Hoadley K, Kushwaha D, Ramakrishnan V, Li S, et al. miR-181d: a predictive glioblastoma biomarker that downregulates MGMT expression. Neuro-oncology. 2012;14(6):712-9. [DOI:10.1093/neuonc/nos089] [PMID] [PMCID]
- Soejima H, Zhao W, Mukai T. Epigenetic silencing of the MGMT gene in cancer. Biochemistry and cell biology. 2005;83(4):429-37. [DOI:10.1139/o05-140] [PMID]
- Osanai T, Takagi Y, Toriya Y, Nakagawa T, Aruga T, Iida S, et al. Inverse correlation between the expression of O 6-methylguanine-DNA methyl transferase (MGMT) and p53 in breast cancer. Japanese journal of clinical oncology. 2005;35(3):121-5. [DOI:10.1093/jjco/hyi036] [PMID]
- Costello JF, Futscher BW, Tano K, Graunke DM, Pieper RO. Graded methylation in the promoter and body of the O6-methylguanine DNA methyltransferase (MGMT) gene correlates with MGMT expression in human glioma cells. Journal of Biological Chemistry. 1994;269(25):17228-37.
- Esteller M, Hamilton SR, Burger PC, Baylin SB, Herman JG. Inactivation of the DNA repair gene O6-methylguanine-DNA methyltransferase by promoter hypermethylation is a common event in primary human neoplasia. Cancer research. 1999;59(4):793-7.
- Esteller M, Risques R-A, Toyota M, Capella G, Moreno V, Peinado MA, et al. Promoter hypermethylation of the DNA repair gene O6-methylguanine-DNA methyltransferase is associated with the presence of G: C to A: T transition mutations in p53 in human colorectal tumorigenesis. Cancer Research. 2001;61(12):4689-92.
- Herfarth KKF, Brent TP, Danam RP, Remack JS, Kodner IJ, Wells SA, et al. A specific CpG methylation pattern of the MGMT promoter region associated with reduced MGMT expression in primary colorectal cancers. Molecular carcinogenesis. 1999;24(2):90-8. https://doi.org/10.1002/(SICI)1098-2744(199902)24:2 3.0.CO;2-B [DOI:10.1002/(SICI)1098- 2744(199902)24:23.0.CO;2-B]
- Stephen J, Chen K, Merritt J, Chitale D, Divine G, Worsham M. Methylation markers differentiate thyroid cancer from benign nodules. Journal of endocrinological investigation. 2018;41(2):163-70. [DOI:10.1007/s40618-017-0702-2] [PMID]
- Santos JC, Bastos AU, Cerutti JM, Ribeiro ML. Correlation of MLH1 and MGMT expression and promoter methylation with genomic instability in patients with thyroid carcinoma. Bmc Cancer. 2013;13(1):79. [DOI:10.1186/1471-2407-13-79] [PMID] [PMCID]
- Gopal E, Fei Y-J, Sugawara M, Miyauchi S, Zhuang L, Martin P, et al. Expression of slc5a8 in kidney and its role in Na+-coupled transport of lactate. Journal of Biological Chemistry. 2004;279(43):44522-32. [DOI:10.1074/jbc.M405365200] [PMID]
- Takebe K, Nio J, Morimatsu M, Karaki S-I, Kuwahara A, Kato I, et al. Histochemical demonstration of a Na+-coupled transporter for short-chain fatty acids (slc5a8) in the intestine and kidney of the mouse. Biomedical Research. 2005;26(5):213-21. [DOI:10.2220/biomedres.26.213] [PMID]
- Iwanaga T, Takebe K, Kato I, Karaki S-I, Kuwahara A. Cellular expression of monocarboxylate transporters (MCT) in the digestive tract of the mouse, rat, and humans, with special reference to slc5a8. Biomedical research. 2006;27(5):243-54. [DOI:10.2220/biomedres.27.243] [PMID]
- Martin PM, Gopal E, Ananth S, Zhuang L, Itagaki S, Prasad BM, et al. Identity of SMCT1 (SLC5A8) as a neuron‐specific Na+‐coupled transporter for active uptake of l‐lactate and ketone bodies in the brain. Journal of neurochemistry. 2006;98(1):279-88. [DOI:10.1111/j.1471-4159.2006.03878.x] [PMID]
- Galrão AL, Camargo RY, Friguglietti CU, Moraes L, Cerutti JM, Serrano-Nascimento C, et al. Hypermethylation of a New Distal Sodium/Iodide Symporter (NIS) Enhancer (NDE) Is Associated With Reduced NIS Expression in Thyroid Tumors. The Journal of Clinical Endocrinology & Metabolism. 2014;99(6):E944-E52. [DOI:10.1210/jc.2013-1450] [PMID]
- Khatami F, Larijani B, Heshmat R, Keshtkar A, Mohammadamoli M, Teimoori-Toolabi L, et al. Meta-analysis of promoter methylation in eight tumor-suppressor genes and its association with the risk of thyroid cancer. PloS one. 2017;12(9):e0184892. [DOI:10.1371/journal.pone.0184892] [PMID] [PMCID]
- Schagdarsurengin U, Gimm O, Dralle H, Hoang-Vu C, Dammann R. CpG island methylation of tumor-related promoters occurs preferentially in undifferentiated carcinoma. Thyroid. 2006;16(7):633-42. [DOI:10.1089/thy.2006.16.633] [PMID]
- Porra V, Ferraro-Peyret C, Durand C, Selmi-Ruby S, Giroud H, Berger-Dutrieux N, et al. Silencing of the tumor suppressor gene SLC5A8 is associated with BRAF mutations in classical papillary thyroid carcinomas. The Journal of Clinical Endocrinology & Metabolism. 2005;90(5):3028-35. [DOI:10.1210/jc.2004-1394] [PMID]
- Schagdarsurengin U, Gimm O, Dralle H, Hoang-Vu C, Dammann R. CpG island methylation of tumor-related promoters occurs preferentially in undifferentiated carcinoma. Thyroid. 2006;16(7):633-42. [DOI:10.1089/thy.2006.16.633] [PMID]
- Pabalan N, Kunjantarachot A, Ruangpratheep C, Jarjanazi H, Christofolini DM, Barbosa CP, et al. Potential of RASSF1A promoter methylation as biomarker for endometrial cancer: A systematic review and meta-analysis. Gynecologic oncology. 2017;146(3):603-8. [DOI:10.1016/j.ygyno.2017.06.017] [PMID]
- Tommasi S, Dammann R, Zhang Z, Wang Y, Liu L, Tsark WM, et al. Tumor susceptibility of Rassf1a knockout mice. Cancer Research. 2005;65(1):92-8.
- Amato E, Barbi S, Fassan M, Luchini C, Vicentini C, Brunelli M, et al. RASSF1 tumor suppressor gene in pancreatic ductal adenocarcinoma: correlation of expression, chromosomal status and epigenetic changes. BMC cancer. 2016;16(1):11. [DOI:10.1186/s12885-016-2048-0] [PMID] [PMCID]
- Vos MD, Ellis CA, Bell A, Birrer MJ, Clark GJ. Ras uses the novel tumor suppressor RASSF1 as an effector to mediate apoptosis. Journal of Biological Chemistry. 2000;275(46):35669-72. [DOI:10.1074/jbc.C000463200] [PMID]
- Xu H, Zhan W, Chen Z. Ras-Association Domain Family 1 Isoform A (RASSF1A) Gene Polymorphism rs1989839 is Associated with Risk and Metastatic Potential of Osteosarcoma in Young Chinese Individuals: A Multi-Center, Case-Control Study. Medical science monitor: international medical journal of experimental and clinical research. 2016;22:4529. [DOI:10.12659/MSM.901994] [PMID] [PMCID]
- Grawenda A, O'neill E. Clinical utility of RASSF1A methylation in human malignancies. British journal of cancer. 2015;113(3):372. [DOI:10.1038/bjc.2015.221] [PMID] [PMCID]
- da Costa Prando É, Cavalli LR, Rainho C. Evidence of epigenetic regulation of the tumor suppressor gene cluster flanking RASSF1 in breast cancer cell lines. Epigenetics. 2011;6(12):1413-24. [DOI:10.4161/epi.6.12.18271] [PMID] [PMCID]
- Shao C, Dai W, Li H, Tang W, Jia S, Wu X, et al. The relationship between RASSF1A gene promoter methylation and the susceptibility and prognosis of melanoma: A meta-analysis and bioinformatics. PloS one. 2017;12(2):e0171676. [DOI:10.1371/journal.pone. 0171676] [PMID] [PMCID]
- Huang Y, Guan H, Liu C, Liu D, Xu B, Jiang L, et al. Association between RASSF1A promoter methylation and renal cell cancer susceptibility: a meta-analysis. Genet Mol Res. 2016;15(April (2)). [DOI:10.4238/gmr.15026994]
- Wen G, Wang H, Zhong Z. Associations of RASSF1A, RARβ, and CDH1 promoter hypermethylation with oral cancer risk: A PRISMA-compliant meta-analysis. Medicine. 2018;97(11). [DOI:10.1097/MD.0000000000009971] [PMID] [PMCID]
- Schagdarsurengin U, Gimm O, Hoang-Vu C, Dralle H, Pfeifer GP, Dammann R. Frequent epigenetic silencing of the CpG island promoter of RASSF1A in thyroid carcinoma. Cancer research. 2002;62(13):3698-701.
- Nakamura N, Carney JA, Jin L, Kajita S, Pallares J, Zhang H, et al. RASSF1A and NORE1A methylation and BRAF V600E mutations in thyroid tumors. Laboratory investigation. 2005;85(9):1065. [DOI:10.1038/labinvest.3700306] [PMID]
- Zhang K, Li C, Liu J, Tang X, Li Z. DNA methylation alterations as therapeutic prospects in thyroid cancer. Journal of endocrinological investigation. 2018:1-8.
- Niu H, Yang J, Yang K, Huang Y. The relationship between RASSF1A promoter methylation and thyroid carcinoma: A meta-analysis of 14 articles and a bioinformatics of 2 databases (PRISMA). Medicine. 2017;96(46). [DOI:10.1097/MD.0000000000008630] [PMID] [PMCID]
- Bai J, Zhang X, Hu K, Liu B, Wang H, Li A, et al. Silencing DNA methyltransferase 1 (DNMT1) inhibits proliferation, metastasis and invasion in ESCC by suppressing methylation of RASSF1A and DAPK. Oncotarget. 2016;7(28):44129-41. [DOI:10.18632 /oncotarget.9866] [PMID] [PMCID]
- Zhang H, He J, Li J, Tian D, Gu L, Zhou M. Methylation of RASSF1A gene promoter is regulated by p53 and DAXX. The FASEB Journal. 2013;27(1):232-42. [DOI:10.1096 /fj.12-215491] [PMID] [PMCID]
- Puto LA, Reed JC. Daxx represses RelB target promoters via DNA methyltransferase recruitment and DNA hypermethylation. Genes & development. 2008;22(8):998-1010. [DOI:10.1101/gad.1632208] [PMID] [PMCID]

