Introduction
The cancer of cervix is ranked as the second most common cancer and a common cause of cancer-related death, particularly in developing countries (1). Cervical cancer spreads to pelvic lymph nodes, leading to a high incidence of cancer recurrence and unfavorable survival rates (2, 3). Advancements in surgical management and chemoradiotherapy could not improve patient outcomes, particularly if pelvic lymph node metastases were found (4). The European Society of Gynecological Oncology guidelines recommend a preoperative evaluation of pelvic lymph nodes for early design of management strategies (5). Therefore, it is urgent to detect new predictive biomarkers for the early assessment of pelvic lymph node status in patients with cervical cancer (6, 7).
Fatty acid binding protein 4 (FABP4) is an intracellular lipid chaperone that can carry fatty acids to many organelles (8). Recent studies have incriminated FABP4 in carcinogenesis and metastatic potential of cancers of many organs such as colon, breast, and ovary (9).
Subunit 2 of the GINS complex (GINS2), which belongs to the GINS complex family, encodes a protein that plays a role in initiating DNA replication and controlling cell cycle and normal cell division. GINS2 expression was found in many cancer types and was associated with carcinogenesis, cancer progression, and metastases (10). Epithelial–mesenchymal transition (EMT) is characterized by a change of malignant cells from epithelial to spindle cells that can invade and metastasize. Chromobox homologue 7 (CBX7) was found to promote cancer occurrence and spread through the promotion of EMT. CBX7 was found to have several functions in cancer according to cancer type (11).
However, the prognostic and predictive functions of FABP4, GINS2, and CBX7 in the cancer cervix and their association with the occurrence of lymph node metastases are not sufficiently clarified.
The current study aimed to assess the expression of FABP4, GINS2, and CBX7 in cancer cervix tissue to detect their prognostic and predictive roles in developing lymph node metastases in cancer cervix patients.
Material and Methods
We included 62 patients with cancer cervix who underwent a radical hysterectomy in addition to lymphadenectomy in the Gynecology and Obstetrics Department and General Surgery Department, Faculty of Medicine, Zagazig University, from 2017 to 2022. The patients were followed to detect disease recurrence and survival rates. Tissue samples were sent to the Pathology Department, Faculty of Medicine, Zagazig University, where they were processed, diagnosed, graded, and staged. Sections from paraffin blocks of prepared samples are stained with FABP4, GINS2, and CBX7 using immunohistochemistry.
Inclusion Criteria
Patients with a sure diagnosis of operable cervical carcinoma, Stages I-III, who accepted to be included in the study.
Exclusion Criteria
Inoperable patients are patients who received preoperative chemotherapy or radiotherapy.
Ethical Approval: The local ethics committee of the Faculty of Medicine of Zagazig University approved the study with an approval code of (ZU-IRB #10140). Written informed consent was obtained from all patients prior to enrolment in the study.
Immunohistochemistry:
Sections from paraffin blocks of included cases were incubated with rabbit monoclonal anti-FABP4 antibody (1:50 dilution; Proteintech, 12802- 1-AP, China), anti-GINS2 antibody (1:600; HPA057285; Sigma) and anti-CBX7 (1:200 dilution; ab2187 (12-17)3; Abcam, Cambridge, UK). Markers expression was evaluated by assessment of intensity and extent of the stain: staining intensity (no = 0, weak = 1, moderate = 2, strong = 3) and extent (no = 0, less than 30% = 1, between 30 and 60% = 2, more than 60% = 3). The final stain index was reached by multiplication of the stain intensity and extent scores, reaching values of 0-9. A stain score ≥4 indicates high expression; a score of <4 indicates low expression.
Statistical Analysis
We performed statistical analyses using Graph Pad software (version 7.0). FABP4, GINS2, and CBX7 expression levels and association with clinicopathological and prognostic parameters were analyzed by the χ2 test. We measured DFS and OS rates using the Kaplan–Meier method and analyzed the differences in survival using the Log-rank test. The univariate and multivariate Cox proportional hazard models tested the predictive values of these ten variables. All statistical tests were two-sided. A P-value< 0.05 was considered statistically significant.
Results
The demographic and clinicopathological parameters of the included patients are summarized in Table 1. The expression of FABP4 expression was upregulated in cervical cancer tissues. Its expression was positively associated with the age of the patient (P=0.002), the size of the large tumor (P=0.004), the high grade (P=0.005), involvement of the lymphatic vascular space, infiltration of para-uterine organs, advanced stage of FIGO, resistance to chemotherapy and tumor recurrence (P<0.001). Patients with high expression of FABP4 had shorter rates of recurrence-free survival (RFS) and overall survival (OS) rates (P<0.001) Figures 1 and 4; Tables 2, 3, 4, 5, and 6.
The expression of GINS2 expression was increased in cervical cancer tissues. Its expression was positively associated with the old age of the patient (P=0.002), large tumor size (P=0.004), high grade (P=0.005), involvement of the lymphovascular space, infiltration of para-uterine organs, advanced stage of FIGO, resistance to chemotherapy, tumor recurrence (P<0.001).
Patients with high expression of GINS2 had shorter rates of recurrence-free survival (RFS) and overall survival (OS) rates (P<0.001) Figures 1 and 4; Tables 2, 3, 4, 5, and 6.
The expression of CBX7 expression was markedly downregulated in cervical cancer tissues. High expression of CBX7 was associated with a low grade (P=0.049), absence of myometrial invasion (P=0.039), absence of lymphovascular space involvement, absence of para-uterine infiltration, early stage of FIGO (P=0.022), response to chemotherapy (P=0.33) and lower incidence of tumor recurrence (P=0.028).
Patients with high CBX7 expression had longer recurrence-free survival (RFS) and overall survival (OS) rates (P<0.001) Figures 3 and 4; Tables 2, 3, 4, 5, and 6.
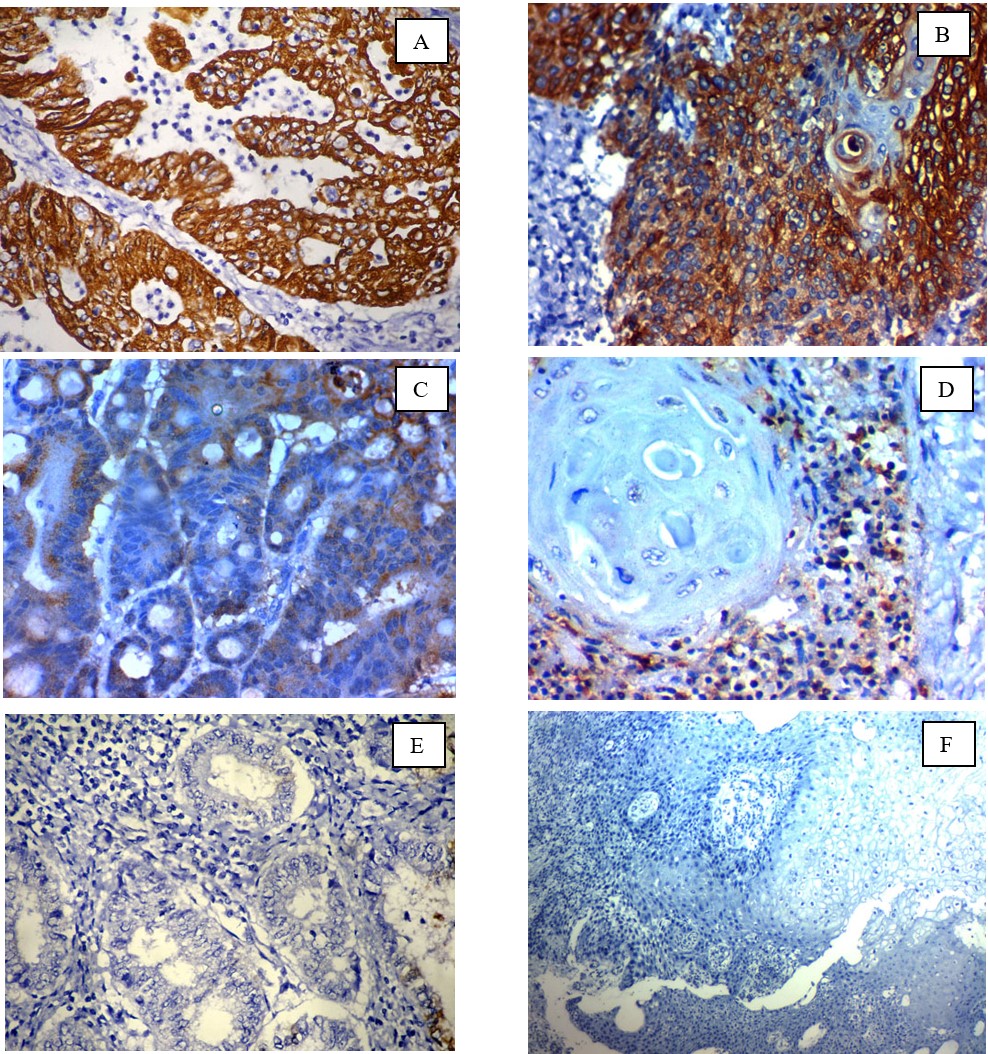
Fig. 1.Immunohistochemical expression of FABP4 in cervical carcinoma: (A); high cytoplasmic expression in high-grade adeno-carcinoma of the cervix ×400, (B); high cytoplasmic expression in high-grade squamous cell carcinoma of the cervix (C); Low cytoplasmic expression in low-grade adeno-carcinoma of the cervix ×400, (D); Low cytoplasmic expression in low-grade squamous cell carcinoma of the cervix (E) ); Negative cytoplasmic expression in low-grade adeno-carcinoma of the cervix ×400, (F) Negative cytoplasmic expression in low-grade squamous cell carcinoma of the cervix x 200
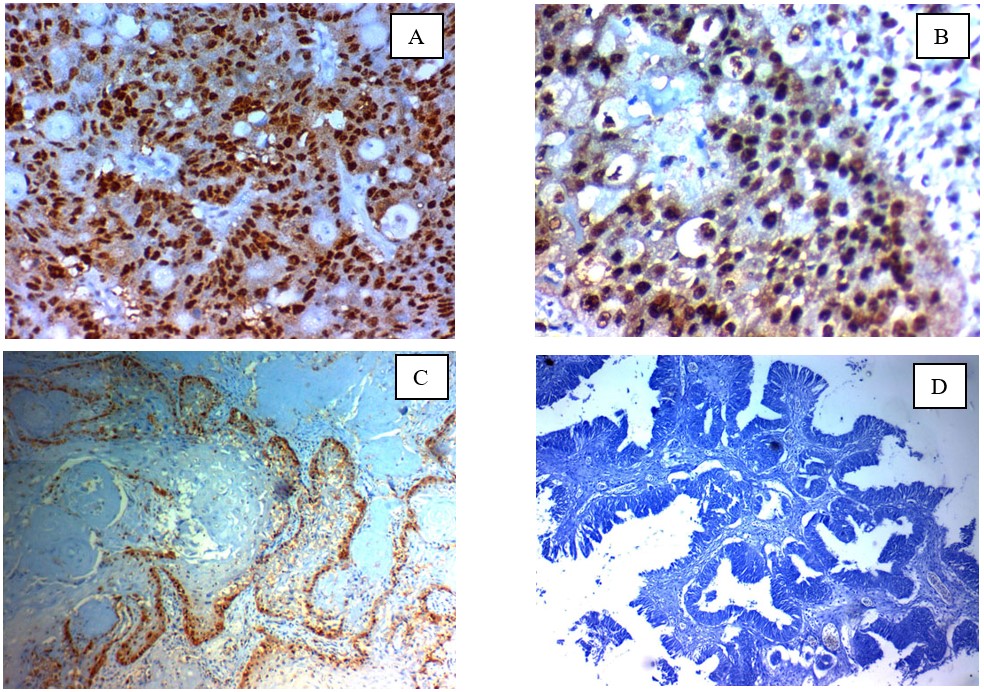
Fig. 2.Immunohistochemical expression of GINS2 in cervical carcinoma: (A); high nuclear expression in high grade adenocarcinoma of the cervix ×400, (B); high nuclear expression in high grade squamous cell carcinoma of the cervix (C); Low nuclear expression in low grade squamous cell carcinoma of the cervix ×400, (D); Negative nuclear expression in low grade adeno-carcinoma of the cervix x 200
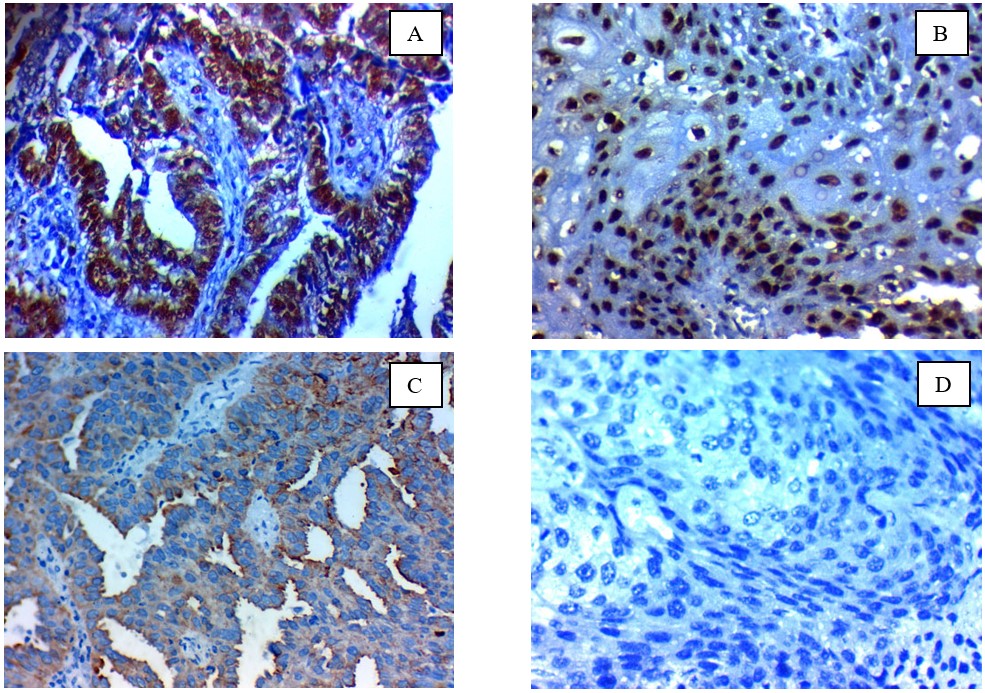
Fig. 3.Immunohistochemical expression of CBX7 in cervical carcinoma: (A); high nuclear expression in low-grade adenocarcinoma of the cervix ×400, (B); high nuclear expression in low-grade squamous cell carcinoma of the cervix (C); Low nuclear expression in high-grade adenocarcinoma of the cervix ×400, (D); Negative nuclear expression in high-grade squamous cell carcinoma of the cervix x 400
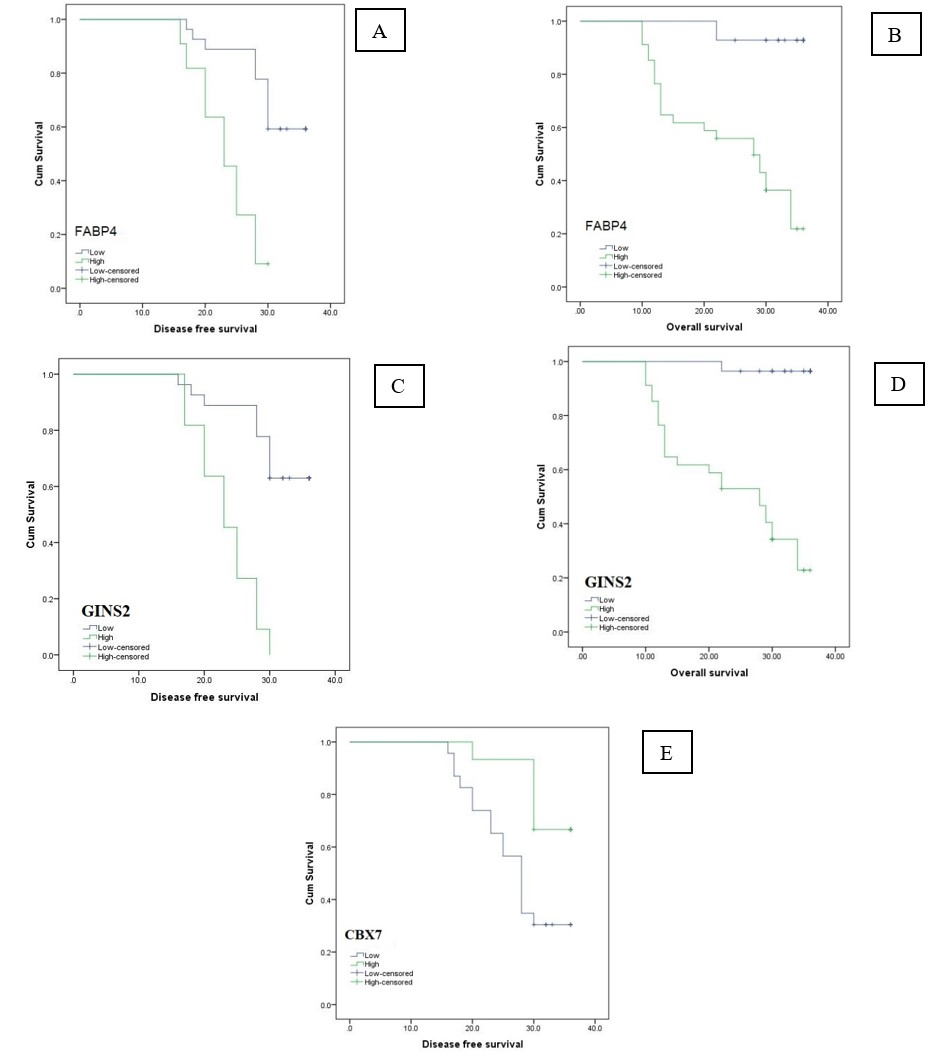
Fig. 4.Kaplan Meir survival curves of DFS and OS rates of the studied cervical carcinoma patients: (A, C, E) DFS rates of the studied cervical carcinoma cases stratified according to FABP4, GINS2 and CBX7 expression respectively, (B, D, F) OS rates of the studied cervical carcinoma cases stratified according to FABP4, GINS2 and CBX7 expression, respectively
| FABP4 | GINS2 | CBX7 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Low | High | P¥ | Low | High | P¥ | Low | High | P¥ | |||
| N=62 | % | N=28 (45.2%) | N=34 (54.8%) | N=28 (45.2%) | N=34 (54.8%) | N=40 (61.3%) | N=22 (38.7%) | ||||
| Age group: | |||||||||||
| ≤ 55 years old | 25 | 40.3 | 26 (92.7) | 11 (32.4) | <0.001* | 16 (57.1) | 9 (26.5) | 0.014* | 9 (22.5) | 16 (72.7) | <0.001* |
| >55 years old | 37 | 59.7 | 2 (7.1) | 23 (67.6) | 12 (42.9) | 25 (73.5) | 31 (77.5) | 6 (27.3) | |||
| Histopatholog: | |||||||||||
| Squamous cell carcinoma | 44 | 71 | 19 (67.9) | 25 (73.5) | 0.78 | 19 (67.9) | 25 (73.5) | 0.78 | 28 (70) | 16 (72.7) | 0.821 |
| Adenocarcinoma | 18 | 29 | 9 (32.1) | 9 (26.5) | 9 (32.1) | 9 (26.5) | 12 (30) | 6 (27.3) | |||
| Size: | |||||||||||
| <4cm | 6 | 9.7 | 6 (21.4) | 0 (0) | 0.006* | 6 (21.4) | 0 (0) | 0.006* | 3 (7.5) | 3 (13.6) | 0.657 |
| ≥4cm | 56 | 90.3 | 22(78.6) | 34 (100) | 22 (78.6) | 34 (100) | 37 (92.5) | 24 (86.4) | |||
| Grade: | |||||||||||
| I | 10 | 16.1 | 10 (35.7) | 0 (0) | <0.001* | 10 (35.7) | 0 (0) | <0.001* | 4 (10) | 6 (27.3) | 0.303 |
| II | 38 | 61.3 | 17 (60.7) | 21 (61.8) | 16 (57.1) | 22 (64.7) | 27 (67.5) | 11 (50) | |||
| III | 14 | 22.6 | 1 (3.6) | 13 (38.2) | 2 (7.1) | 12 (35.3) | 9 (22.5) | 5 (22.7) | |||
| LVSI: | |||||||||||
| Absent | 44 | 71 | 27 (96.4) | 17 (50) | <0.001* | 26 (92.9) | 18 (52.9) | 0.001* | 28 (70) | 16 (72.7) | 0.821 |
| Present | 18 | 29 | 1 (3.6) | 17 (50) | 2 (7.1) | 16 (47.1) | 12 (30) | 6 (27.3) | |||
| Lymph node: | |||||||||||
| Absent | 30 | 48.4 | 22 (78.6) | 8 (23.5) | <0.001* | 22 (78.6) | 8 (23.5) | 0.002* | 17 (42.5) | 13 (59.1) | 0.211 |
| Present | 32 | 51.6 | 6 (21.4) | 26 (76.5) | 6 (21.4) | 26 (76.5) | 23 (57.5) | 9 (40.9) | |||
| Distant metastasis: | |||||||||||
| Absent | 46 | 74.2 | 27 (96.4) | 19 (55.9) | <0.001* | 20 (71.4) | 10 (29.4) | 0.003* | 29 (72.5) | 17 (77.3) | 0.681 |
| Present | 16 | 25.8 | 1 (3.6) | 15 (44.1) | 8 (28.6) | 24 (70.6) | 11 (27.5) | 5 (27.7) | |||
| Stage: | |||||||||||
| I | 6 | 9.7 | 6 (21.4) | 0 (0) | <0.001* | 6 (21.4) | 0 (0) | 0.001* | 3 (7.5) | 3 (13.6) | 0.286 |
| II | 24 | 38.7 | 16 (57.1) | 8 (23.5) | 14 (50) | 10 (29.4) | 14 (35) | 10 (45.5) | |||
| III | 16 | 25.8 | 5 (17.9) | 11 (32.4) | 6 (21.4) | 10 (29.4) | 12 (30) | 4 (18.2) | |||
| IV | 16 | 25.8 | 1 (3.6) | 15 (44.1) | 2 (7.1) | 14 (41.2) | 11 (27.5) | 5 (22.7) | |||
| N = 62 | % | |
|---|---|---|
| Treatment: | ||
| SurgerySurgery and radiotherapySurgery and chemotherapySurgery, radiotherapy, and chemotherapyRadiotherapyChemotherapy | 1310171444 | 2116.127.422.66.56.5 |
| Response: | ||
| PDSDPRCR | 374714 | 59.76.511.322.6 |
| Response: | ||
| OARNR | 4121 | 66.133.9 |
| Outcome: | ||
| AliveDead | 3725 | 59.740.3 |
| Disease free survival (months) (N=38): | ||
| Mean ± SDRange | 29.03 ± 6.4916 - 36 | |
| Overall survival (months): | ||
| Mean ± SDRange | 27.68 ± 9.1810 – 36 | |
| FABP4 | GINS2 | CBX7 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Low | High | P | Low | High | P | Low | High | P | |
| N=28(45.2%) | N=34(54.8%) | N=28(45.2%) | N=34 (54.8%) | N=40 (64.5%) | N=22((35.5%) | ||||
| Treatment response: | |||||||||
| CR | 27 (96.4) | 10 (29.4) | <0.001* | 26 (92.9) | 11(32.4) | <0.001* | 32 (57.5) | 14 (63.6) | 0.933 |
| PR | 1 (3.6) | 3 (8.8) | 2 (7.1) | 2 (5.9) | 3 (7.5) | 1 (4.5) | |||
| SD | 0 (0) | 7 (20.6) | 0 (0) | 7 (20.6) | 4 (10) | 3 (13.6) | |||
| PD | 0 (0) | 14 (41.2) | 0 (0) | 14(41.2) | 10 (25) | 4 (18.2) | |||
| Response: | |||||||||
| OAR | 28 (100) | 13(38.2) | <0.001* | 28 (100) | 13(38.2) | <0.001* | 26 (65) | 15(68.2) | 0.8 |
| NR | 0 (0) | 25(61.8) | 0 (0) | 21(61.8) | 14 (35) | 7 (31.8) | |||
| Recurrence (n=38): | |||||||||
| Absent | 16(59.3) | 1 (9.1) | <0.001* | 17 (63) | 0 (0) | <0.001* | 7 (30.4) | 10(66.7) | 0.028* |
| Present | 11(40.7) | 10(90.9) | 10 (37) | 11 (100) | 16(69.6) | 5 (33.3) | |||
| FABP4 | GINS2 | CBX7 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Low | High | P | Low | High | P | Low | High | P | |
| N=28 (45.2%) | N=34 (54.8%) | N=28 (45.2%) | N=34 (54.8%) | N=38 (61.3%) | N=24 (38.7%) | ||||
| Treatment response: | |||||||||
| CR | 27 (96.4) | 10 (29.4) | <0.001* | 26 (92.9) | 11 (32.4) | <0.001* | 31 (81.6) | 6 (25) | <0.001* |
| PR | 1 (3.6) | 3 (8.8) | 2 (7.1) | 2 (5.9) | 4 (10.5) | 0 (0) | |||
| SD | 0 (0) | 7 (20.6) | 0 (0) | 7 (20.6) | 2 (5.3) | 5 (20.8) | |||
| PD | 0 (0) | 14 (41.2) | 0 (0) | 14 (41.2) | 1 (2.6) | 13 (54.2) | |||
| Response: | |||||||||
| OAR | 28 (100) | 13 (38.2) | <0.001* | 28 (100 | 13 (38.2) | <0.001* | 35 (92.1) | 6 (25) | <0.001* |
| NR | 0 (0) | 25 (61.8) | 0 (0) | 21 (61.8) | 3 (7.9) | 18 (75) | |||
| Recurrence (n=38): | |||||||||
| Absent | 16 (59.3) | 1 (9.1) | <0.001* | 17 (63) | 0 (0) | <0.001* | 17 (53.1) | 0 (0) | <0.001* |
| Present | 11 (40.7) | 10 (90.9) | 10 (37) | 11 (100) | 15 (46.9) | 6 (100) | |||
| Disease free survival: | |||||||||
| Mean ± SD | 31.41±5.62 | 23.18 ± 4.58 | <0.001*∞ | 33.69 ± 3.4 | 29.36±5.7 | <0.001*∞ | 32.88±4.6 | 31.33±6.81 | 0.003*∞ |
| Range | 17 - 36 | 16 - 30 | 22 - 36 | 16 - 36 | 20 - 36 | 30 - 34 | |||
| Overall survival: | |||||||||
| Median | 36 | 28 | <0.001*# | 32 | 23 | <0.001*# | 30 | 25 | <0.001*# |
| Range | 22 - 36 | 10 - 36 | 16 - 36 | 17 - 30 | 16 - 36 | 23 - 28 | |||
| FABP4 | GINS2 | CBX7 | ||||
|---|---|---|---|---|---|---|
| Phi | P | Phi | P | Phi | P | |
| FABP4 | 0.87 | <0.001* | 0.721 | <0.00* | ||
| GINS2 | 0.87 | <0.001* | 0.721 | <0.001* | ||
| CBX7 | 0.721 | <0.001* | 0.721 | <0.001* | ||
| Patients’ outcome | FABP4 | GINS2 | CBX7 | |||
|---|---|---|---|---|---|---|
| High | Low | High | Low | High | Low | |
| N=34 (%) | N=28 (%) | N=34 (%) | N=28 (%) | N=22 (%) | N=40 (%) | |
| Outcome:DeadAlive | 23 (67.6)11 (32.4) | 2 (7.1)26 (92.9) | 24 (70.6)10 (29.4) | 1 (3.6)27 (96.4) | 18 (81.8)4 (18.2) | 19 (47.5)21 (52.5) |
| P | <0.001** | <0.001** | <0.008* | |||
| Odds ratio | 27.18 | 64.8 | 4.97 | |||
| 95% confidence interval | 5.45 – 136.68 | 7.72 – 544.14 | 1.43 – 17.34 | |||
Discussion
The current study showed that high FABP4 expression in cervical cancer tissues is associated with unfavorable prognostic parameters and poor outcomes. Furthermore, we showed that increased expression of FABP4 is associated with a higher incidence of lymph node metastases. Similarly, Guoqing Li et al. (6) showed that FABP4 is associated with the development of lymph node metastases and a poor prognosis in patients with cervical cancer. Our results are important because lymph node status is essential in preparing and assigning treatment in patients with cancer cervix.
Furthermore, Guoqing Li et al. (6) showed that loss of FABP4 expression inhibits cervical cancer cell proliferation, invasion, and metastases. Taken together, FABP4 increased the malignant potential of cervical cancer cells. Thus, it could be a significant prognostic and predictive biomarker of lymph node metastases in cancer cervix patients (6).
It was recently found that there are many predictive risk factors for lymph node metastases in the cancer cervix, such as the advanced FIGO stage, the large size of the tumor, the depth of cervical stromal invasion, and lymph-vascular invasion (18-20).
We showed that FABP4 was an essential predictive risk factor for lymph node metastases in cancer cervix patients, like Guoqing Li et al. (6), who hypothesized that the preoperative evaluation of FABP4 expression levels in colposcopy biopsy tissues could provide clinicians with key clues for better surgical and chemotherapeutic management.
Furthermore, we demonstrated a positive association between FABP4 expression and a poor OS rate; therefore, FABP4 might be a beneficial predictive and prognostic biomarker for cancer patients with cancer cervix. In addition to other studies, our findings indicate that more aggressive and individualized management strategies are needed to manage patients with FABP4 overexpression (6).
The occurrence of lymph node metastases is a complex process controlled by many genes that increase the invasiveness of cancer cells (21). According to Guoqing Li et al. (6), FABP4 has a role in controlling epithelial markers, and its over-expression could increase mesenchymal markers, facilitating the metastatic process. Moreover, it was observed that specific inhibitors of FABP4 were found to decrease cancer progression by reducing proliferation, invasive ability, and metastasis of cancer cells. Furthermore, FABP4 enhances cancer progression by controlling lipid metabolism, the AKT pathway, and stimulating EMT (22).
According to Zhang et al., the expression of FABP4 in CRC tissues is positively associated with cancer progression and lymph node metastases (23). Luo et al. (24) revealed a similar role in patients with pancreatic adenocarcinoma tissues.
In contrast to our results, Zhong et al. (25) reported an inverse association between FABP4 expression levels, proliferative and invasive liability of hepatocellular carcinoma cells, and its low expression levels related to unfavorable survival rates (25). Different roles in different types of cancer may explain these results. Therefore, more studies are needed to clarify the precision of FABP4 in the cancer cervix.
We evaluated another novel expression of prognostic markers that has not been markedly evaluated in cervical cancer, GINS2. We showed that GINS2 is upregulated in cervical cancer tissues, associated with well-established poor prognostic parameters such as high-grade deep stromal invasion, and positively correlated with poor survival and unfavorable outcomes. Our results were in line with Fei Ouyang et al. (7), who reported a high expression of GINS2 in cervical cancer tissues and an association with unfavorable outcomes. Moreover, they showed that down-regulation of GINS2 down-regulation reduces cancer cells, proliferation, invasion, and metastases; thus, it has a role in the progression of cancer cervix.
A similar prognostic role of GINS2 up-regulation is present in other cancers, such as lung, breast, and cholangiocarcinoma (26, 27). Furthermore, high expression of GINS2 was related to cancer stem cell activation and progression in breast cancer (10). Our results indicated that GINS2 plays many roles in cancer oncogenesis and progression.
Many mechanisms explain the prognostic roles of GINS2 in cancer, such as cancer stem cell activation, inhibition of apoptosis, and cell cycle control. In cancer cervix, evaluation of the status of lymph nodes is needed preoperatively for an accurate designation of treatment strategy (28). Performing lymphadenectomy in early-stage patients without lymph node metastases leads to morbidity and postoperative complications (29). There is no accurate predictive biomarker or imaging technique for preoperative diagnosis of lymph node metastases; also, sentinel lymph node biopsy could not accurately assess its occurrence (30, 31).
According to our findings, GINS2 overexpression was positively correlated with lymph node metastases and aggressive cancer cervix; therefore, it could be considered a novel therapeutic target and prognostic marker.
We showed that both FABP4 and GISN2 are upregulated in cancer cervix tissues and are related to unfavorable outcomes.
We evaluated the expression of another biomarker (CBX7) in cervical cancer tissues to confirm the role of these novel biomarkers.
Our results indicated that CBX7 negative expression was associated with poor prognostic parameters and unfavorable outcomes in cervical cancer patients. Similarly, the results of Ping Tian et al. and Ramirez et al. (11, 32) showed that the absence of CBX7 in cancer cervix tissues was related to unfavorable prognostic parameters, such as the presence of lymph nodes and blood metastases, to unfavorable survival outcomes.
Our results were in line with previous results in other cancers, such as glioma and pancreatic carcinoma (33, 34).
Ping Tian et al. (11) explained the oncogenic functions of loss of CBX7 by reducing levels of E-cadherin in tumor cells, thus increasing invasion and metastases by losing epithelial characteristics and having a mesenchymal phenotype that promotes the progression of different types of cancers by the initiation of EMT (35, 36). Unlike our results, previous studies by Federico et al. (37), Sepe et al. (38), and Shinjo et al. (39) showed that CBX7 might play an oncogenic role in cancer progression in some cancers, such as ovarian and colon cancer.
We showed a positive association between FABP4 and GISN2 expression levels in cancer cervix tissues, and both were inversely associated with the expression of CBX7.
Conclusion
We reported prognostic roles of FABP4, GISN2, and CBX7 in cervical cancer tissues, and we demonstrated oncogenic roles of FABP4 and GISN2 in addition to onco-suppressive roles of CBX7 in cervical cancer. Our results indicated that FABP4, GISN2, and CBX7 might be considered predictive biomarkers to the occurrence of lymph node metastases in the cancer cervix preoperatively, which could be beneficial in accurate preoperative designing therapy.
We evaluated the expression of novel biomarkers not extensively studied in cervical cancer. Furthermore, we assessed the expression of these markers in different grades and stages of cancer cervix patients.
Although our research highlighted the important aim of gynecologic surgeons by evaluating novel predictive biomarkers of lymph node metastases in cancer cervix preoperatively, there are some limitations, such as small sample size and using only immunohistochemistry for tissue protein expression of the biomarkers. To validate our findings, we recommend including a larger cohort of patients with cervical cancer patients; additionally, a molecular evaluation of the markers is needed to prove our findings.
Conflict of Interest
None.
Acknowledgments
None.
References
- Fitzmaurice C, Allen C, Barber RM, Barregard L, Bhutta ZA, Brenner H. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA Oncol. 2017; 3(4):524-48.
- Hong K-S, Ju W, Choi HJ, Kim JK, Kim M-h. Differential diagnostic performance of magnetic resonance imaging in the detection of lymph node metastases according to the tumor size in early-stage cervical cancer patients. Int J Gynecol Cancer. 2010; 20:5.
- Meva J, Chaudhary RK, Bhaduri D, Bhatia M, Hatti S, Ba RJIJoGC. Lacunae in International Federation of Gynecology and Obstetrics (FIGO) classification for cervical carcinoma: observational study using TNM classification as comparator. Int J Gynecol Cancer. 2013; 23:6.
- Cibula D, Dostalek L, Hillemanns P, Scambia G, Jarkovsky J, Persson J. Completion of radical hysterectomy does not improve survival of patients with cervical cancer and intraoperatively detected lymph node involvement: ABRAX international retrospective cohort study. Euro J Cancer.. 2021; 143:88-100.
- Cibula D, Pötter R, Planchamp F, Avall-Lundqvist E, Fischerova D, Haie-Meder C. The European Society of Gynaecological Oncology/European Society for Radiotherapy and Oncology/European Society of Pathology guidelines for the management of patients with cervical cancer. Virchows Archiv.. 2018; 472:919-36.
- Li G, Wu Q, Gong L, Xu X, Cai J, Xu L. FABP4 is an independent risk factor for lymph node metastasis and poor prognosis in patients with cervical cancer. Cancer Cell Int. 2021; 21(1):1-5.
- Ouyang F, Liu J, Xia M, Lin C, Wu X, Ye L. GINS2 is a novel prognostic biomarker and promotes tumor progression in early-stage cervical cancer Corrigendum in/10 3892/or 2021 8016. Oncol Rep. 2017; 37(5):2652-62.
- Zimmerman AW, Veerkamp JH. New insights into the structure and function of fatty acid-binding proteins. Cell Mol Life Sci.. 2002; 59:1096-116.
- Tian W, Zhang W, Zhang Y, Zhu T, Hua Y, Li H. FABP4 promotes invasion and metastasis of colon cancer by regulating fatty acid transport. Cancer Cell Int.. 2020; 20:1-3.
- Zheng M, Zhou Y, Yang X, Tang J, Wei D, Zhang Y. High GINS2 transcript level predicts poor prognosis and correlates with high histological grade and endocrine therapy resistance through mammary cancer stem cells in breast cancer patients. Breast Cancer Res and Treat.. 2014; 148:423-36.
- Tian P, Zhang C, Ma C, Ding L, Tao N, Ning L. Decreased chromobox homologue 7 expression is associated with epithelial-mesenchymal transition and poor prognosis in cervical cancer. Open Med. 2021; 16(1):410-8.
- Ahmed MM, Gebriel MG, Morad EA, Saber IM, Elwan A, Salah M. Expression of immune checkpoint regulators, cytotoxic T-lymphocyte antigen-4, and programmed death-ligand 1 in Epstein-Barr virus-associated nasopharyngeal carcinoma. Appl Immunohistochem Mol Morphol. 2021; 29(6):401-8.
- Alabiad MA, Elderey MS, Shalaby AM, Nosery Y, Gobran MAJAI, Morphology M. The Usefulness of 4 Immunoperoxidase Stains Applied to Urinary Cytology Samples in the Pathologic Stage of Urothelial Carcinoma: A Study With Histologic Correlation. Appl Immunohistochem Mol Morphol. 2021; 29(6):422-32.
- Elsalam SA, Mansor AE, Sarhan MH, Shalaby AM, Gobran MA, Alabiad MAJIJoGP. Evaluation of apoptosis, proliferation, and adhesion molecule expression in trophoblastic tissue of women with recurrent spontaneous abortion and infected with Toxoplasma gondii. Int J Gynecol Pathol. 2021; 40(2):124-33.
- Alabiad MA, Harb OA, Taha HF, El Shafaay BS, Gertallah LM, Salama NJP. Prognostic and clinic-pathological significances of SCF and COX-2 expression in inflammatory and malignant prostatic lesions. Pathol Oncol Res.. 2019; 25:611-24.
- Tawfeek SE, Shalaby AM, Alabiad MA, Albackoosh A-AAA, Albakoush KMM, Omira MMAJT. Metanil yellow promotes oxidative stress, astrogliosis, and apoptosis in the cerebellar cortex of adult male rat with possible protective effect of scutellarin: a histological and immunohistochemical study. Tissue Cell.. 2021; 73:101624.
- Alabiad MA, Harb OA, Hefzi N, Ahmed RZ, Osman G, Shalaby AM. Prognostic and clinicopathological significance of TMEFF2, SMOC-2, and SOX17 expression in endometrial carcinoma. Exp Mol Pathol.. 2021; 122:104670.
- Widschwendter P, Janni W, Scholz C, De Gregorio A, De Gregorio N, Friedl TWJAoG. Prognostic factors for and pattern of lymph-node involvement in patients with operable cervical cancer. Arch Gynecol Obstet.. 2019; 300:1709-18.
- Nanthamongkolkul K, Hanprasertpong JJOr. Predictive factors of pelvic lymph node metastasis in early-stage cervical cancer. Oncol Res Treat. 2018; 41(4):194-8.
- Cai J, He X, Wang H, Dong W, Zhang Y, Zhao J. Topographic distribution of lymph node metastasis in patients with stage IB1 cervical cancer: an analysis of 8314 lymph nodes. Radiat Oncol. 2021; 16(1):1-0.
- Bahrami A, Hasanzadeh M, Shahidsales S, Farazestanian M, Hassanian SM, Moetamani Ahmadi M. Genetic susceptibility in cervical cancer: from bench to bedside. J Cell Physiol. 2018; 233(3):1929-39.
- Guaita‐Esteruelas S, Bosquet A, Saavedra P, Guma J, Girona J, Lam EWF. Exogenous FABP4 increases breast cancer cell proliferation and activates the expression of fatty acid transport proteins. Mol Carcinogen. 2017; 56(1):208-17.
- Zhang Y, Zhang W, Min X, Zhujun X, Fangmei A, Qiang Z. High expression of FABP4 in colorectal cancer and its clinical significance. J Zhejiang Univ Sci B.. 2021; 22:136-45.
- Luo Y, Yang Z, Li D, Liu Z, Yang L, Zou Q. LDHB and FABP4 are associated with progression and poor prognosis of pancreatic ductal adenocarcinomas. Appl Immunohistochem Mol Morphol. 2017 May 1; 25(5):351-7.
- Zhong CQ, Zhang XP, Ma N, Zhang EB, Li JJ, Jiang YB. FABP4 suppresses proliferation and invasion of hepatocellular carcinoma cells and predicts a poor prognosis for hepatocellular carcinoma. Cancer Med. 2018; 7(6):2629-40.
- Liu M, Pan H, Zhang F, Zhang Y, Zhang Y, Xia H. Identification of TNM stage-specific genes in lung adenocarcinoma by genome-wide expression profiling. Oncol Lett. 2013; 6(3):763-8.
- Tane S, Sakai Y, Hokka D, Okuma H, Ogawa H, Tanaka Y. Significant role of Psf3 expression in non‐small‐cell lung cancer. Cancer Sci. 2015; 106(11):1625-34.
- Selman TJ, Mann C, Zamora J, Appleyard T-L, Khan KJC. Diagnostic accuracy of tests for lymph node status in primary cervical cancer: a systematic review and meta-analysis. Can Med Assoc J. 2008; 178(7):855-62.
- Matsuura Y, Kawagoe T, Toki N, Tanaka M, Kashimura MJIJoGC. Long-standing complications after treatment for cancer of the uterine cervix-clinical significance of medical examination at 5 years after treatment. Int J Gynecol Cancer. 2006; 16:1.
- Altgassen C, Hertel H, Brandstädt A, Köhler C, Dürst M, Schneider AJJoCO. Multicenter validation study of the sentinel lymph node concept in cervical cancer: AGO Study Group. J Clin Oncol. 2008; 26(18):2943-51.
- Sláma J, Dundr P, Dusek L, Cibula DJGo. High false negative rate of frozen section examination of sentinel lymph nodes in patients with cervical cancer. Gynecol Oncol. 2013; 129(2):384-8.
- Ramirez PT, Frumovitz M, Pareja R, Lopez A, Vieira M, Ribeiro R. Minimally invasive versus abdominal radical hysterectomy for cervical cancer. New Eng J Med. 2018; 379(20):1895-904.
- Yu T, Wu Y, Hu Q, Zhang J, Nie E, Wu W. CBX7 is a glioma prognostic marker and induces G1/S arrest via the silencing of CCNE1. Oncotarget. 2017; 8(16):26637.
- Ni S-J, Zhao L-Q, Wang X-F, Wu Z-H, Hua R-X, Wan C-H. CBX7 regulates stem cell-like properties of gastric cancer cells via p16 and AKT-NF-κB-miR-21 pathways. J Hematol Oncol.. 2018; 11:1-4.
- Bao Z, Xu X, Liu Y, Chao H, Lin C, Li Z. CBX7 negatively regulates migration and invasion in glioma via Wnt/β-catenin pathway inactivation. Oncotarget. 2017; 8(24):39048.
- Wang H, Liu W, Black S, Turner O, Daniel JM, Dean-Colomb W. Kaiso, a transcriptional repressor, promotes cell migration and invasion of prostate cancer cells through regulation of miR-31 expression. Oncotarget. 2016; 7(5):5677.
- Federico A, Sepe R, Cozzolino F, Piccolo C, Iannone C, Iacobucci I. The complex CBX7-PRMT1 has a critical role in regulating E-cadherin gene expression and cell migration. Biochimica et Biophysica Acta (BBA)-Gene Regulatory Mechanisms. 2019 ; 1862(4):509-21.
- Sepe R, Formisano U, Federico A, Forzati F, Bastos AU, D'Angelo D. CBX7 and HMGA1b proteins act in opposite way on the regulation of the SPP1 gene expression. Oncotarget. 2015; 6(5):2680.
- Shinjo K, Yamashita Y, Yamamoto E, Akatsuka S, Uno N, Kamiya A. Expression of chromobox homolog 7 (CBX7) is associated with poor prognosis in ovarian clear cell adenocarcinoma via TRAIL‐induced apoptotic pathway regulation. Int J Cancer. 2014; 135(2):308-18.

