Document Type : Original Research
Authors
1 Department of Pathology, Faculty of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran
2 Department of Hematology and Blood Bank, Cancer Molecular Pathology Research Center, Faculty of Medicine, Mashhad University of Medical Sciences, Mashhad, Iran
Highlights
✅Although multiple studies are strongly suggestive of a role for EBV and HHV-8 in the development of IPF, there was no statistically significant difference in the prevalence of EBV and HHV-8 DNA in the IPF specimens and controls in this study.
Keywords
Subjects
Idiopathic pulmonary fibrosis (IPF) is a chronic debilitating lung disease with unknown etiology, which due to unusual repair mechanisms leads to fibroblastic proliferation and excess collagen deposition (1). Studies show that IPF incidence in primary care is on the rise in the UK with more than 5000 new cases diagnosed annually and a considerable related death toll (2). So far, numerous studies have been performed to identify the possible etiologic factors. Several of these studies have suggested the possible role of certain viral agents in IPF (3-7). Vannella et al. recommended viruses as co-factors responsible for fibrosis progression. They highlighted the role of specific viruses in disease progression in horses and murine models (3). Tang et al. study showed that CMV, EBV, and HHV-8 could be found more frequently in IPF patients compared to the control group and they supported the hypothesis that chronic antigenic stimulation is needed for IPF (6). On the contrary, there is considerable number of studies that could not find any association with those viruses (8-9). Wangoo et al. studied the presence of EBV in lung samples of IPF patients and despite faint immunohistochemical results, they could not find neither DNA nor RNA of the virus (8). Another study by Zamò et al. also could not find any of the viruses they were looking for (EBV, HHV-8) in IPF lung samples using immunohistochemical and molecular techniques (9). Regarding the results of the previous studies and considering the fact that confirmation of this association can be of great importance in treatment plans that would justify the use of antiviral agents to prevent and control this fatal disease, this study was designed to investigate the incidence of Epstein - Barr virus (EBV) and Human Herpesvirus 8 (HHV-8) DNA in lung tissue biopsies with the confirmed diagnosis of IPF and compare the results with the control group.
This is a case-control study performed on formalin-fixed paraffin-embedded (FFPE) tissue samples of lung specimens surgically biopsied at Ghaem Hospital, Mashhad, Iran between 2013 and 2016. Based on previous studies, the sample size for EBV and HHV-8 evaluation was twenty-six (10) and nine (6), respectively. However, we increased the sample size of both the case and control groups up to twenty-nine. The control group was selected from the age and sex-matched normal lung tissue. The exclusion criteria for both groups were inadequacy of tissue for DNA extraction, bad quality of extracted DNA, negative samples in internal control (beta-actin) PCR, and inability to confirm the diagnosis of IPF in the case group. H&E stained slides were retrieved from the archive of pathology department and reevaluated by two expert pathologists to confirm the diagnosis according to the ATS/ERS/JRS/ALAT statement (11). After DNA extraction, twenty- nine samples of the case group and twenty-nine samples of the control group were suitable for polymerase chain reaction (PCR). PCR for EBV and HHV-8 were performed by AmpliSens® EBV PCR kit (Russia) and DNA-Technology, JSC, Kashirskoeshosse (Russia), respectively. PCR product size for EBV and HHV-8 gene were 210, and 265 bp, respectively, and the PCR product size for internal control gene (beta-actin) was 597 bp. Statistical analysis of data was performed using SPSS 16 (SPSS Inc., Chicago, IL. USA) and P-values less than 0.05 were identified as statistically significant.
A total of 58 biopsies, comprised of 29 cases and 29 sex and age-matched controls were included in this study. The mean and standard deviation of age for the case and control groups were 58±7 and 57±9 years, respectively and ranged from 47-74 years. Case group consisted of 16 (55.2%) males and 13 (44.8%) females. Control group consisted of 15(51.7%) male and 14(48.3%) female patients.
Six (20.7%) of the case subjects were positive for EBV DNA, while only one (3.4%) of the control subjects was positive, which was statistically insignificant (P=0.102) (Figure 1).
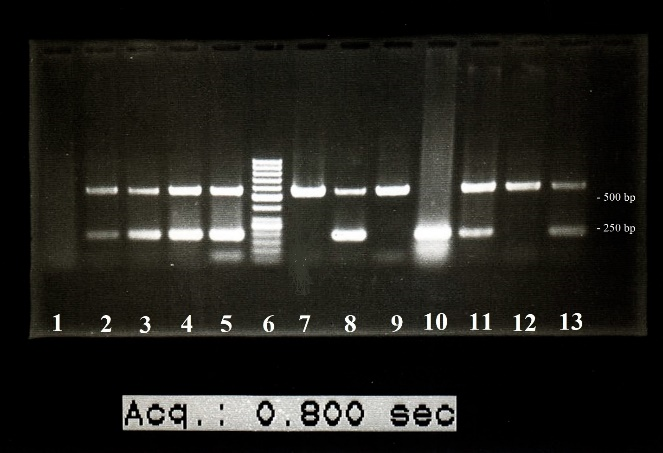
Fig. 1. EBV PCR results: The lanes number 2, 3, 4, 5, 8, 11, and 13 show EBV DNA amplification. Lane number 6 is DNA size marker. Lane number 8 is the positive control. PCR product size for EBV was 210 bp, and the PCR product size for internal control gene (beta-actin) was 597 bp.
As for HHV-8 DNA, the DNA was not detected in any of the control subjects but was found in three (10.3%) of the case subjects. However, the difference was statistically insignificant (P=0.227) (Figure 2).
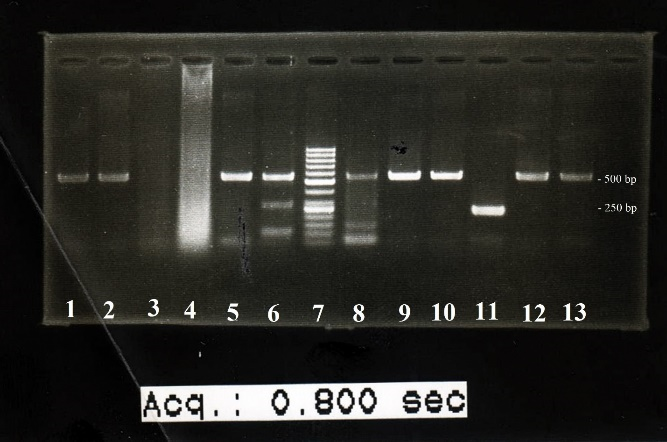
Fig. 2. HHV-8 PCR results: The lane number 6 shows HHV-8 DNA amplification. Lane number 7 is DNA size marker. Lane number 11 is the positive control. PCR product size for HHV-8 gene was 265 bp, and the PCR product size for internal control gene (beta-actin) was 597 bp.
Idiopathic pulmonary fibrosis (IPF) is the most common interstitial lung disease (12), affecting 46 Americans out of 100,000 (13). Unfortunately, the prognosis is poor and the mean survival time from the time of diagnosis is 2–4 years. The only treatment to date is lung transplantation. Although major advances have been made in understanding its pathogenesis, the exact triggers are yet to be known. Numerous studies evaluated the association of cigarette smoking, drugs, gastrointestinal reflux disease, genetic factors, bacterial, and viral infections with IPF. None of these studies could find a definite etiologic factor and the etiology remains unknown. The potential role of viruses has been implicated in disease pathogenesis for different reasons: 1) many of the patients recall a recent viral infection prior to the onset of the disease, and 2) there are some studies revealing that antiviral drugs can reduce lung fibrosis (14-15). One of the first studies on this association compared the serological tests of patients with IPF and those with fibrosis of known cause and showed elevated levels of EBV-specific antibodies (IgA, IgG) in the first group (16). Gammaherpesviruses like EBV exist in pulmonary epithelial cells, B cells and macrophages and their presence induce host response in the form of mild chronic inflammation (17). This level of inflammation can lead to progressive fibrosis in genetically susceptible patients with dysfunctional repair mechanisms. Moreover, increased expression of TGF-β, a key pro-fibrotic mediator was shown in in vitro infection of type II pneumocytes with EBV (18-19). Some studies recommended viruses as co-factors for progression of fibrosis and even showed the role of viruses in disease progression in animal models (3). Even chronic antigenic stimulation was supported in some studies for IPF as they found CMV, EBV, and HHV-8 more frequently in IPF patients compared to the control group (6). Pulkkinen et al. could find the Herpesvirus DNA by using the so-called novel methods such as multiplex PCR-and microarray-based method (7). On the contrary, there are some studies that failed to find EBV and HHV-8 DNA in IPF specimens (8-9). Wangoo et al. could not find the protein, RNA, or DNA of EBV in the lung specimens of IPF patients (8). Likewise, Zamò et al. were unable to identify any evidence supporting the presence of EBV or HHV-8 in IPF lung patients, despite using sensitive methods such as immunohistochemistry, in situ hybridization, and PCR (9). These contradictory results could be attributed to some preanalytic and analytic factors such as the difference in sample size, the fixative, paraffin related issues and the sensitivity of the methods.
As the results of previous studies were contradictory, we decided to study the association of EBV and HHV-8 with IPF in the IPF tissue bank of our institute and compare the results with the controls by using sensitive methods on properly fixed paraffin embedded samples. The results of the present study showed that there is no difference in the prevalence of these two viruses in the studied IPF and control groups. Stewart et al. evaluated the presence of EBV in 27 IPF specimens, and 28 controls by PCR. Thirteen (44%) of IPF specimens and four (14%) of the control group were positive for EBV DNA (P=0.0007). Tsukamoto et al. performed a similar study and found EBV DNA by PCR method in (24/25) 96% of IPF specimens and (10/14) 71% of the control group (P=0.047) (20). In contrast to the present study, these results show a significant relationship between EBV and IPF. However, there are other studies to date that like this study have failed to find EBV DNA in IPF specimens (8-9). One of these recent studies that failed to find a causative role was conducted by researchers who quantified viral RNA expression by employing RNA-seq for 740 viruses in 21 IPF patient lung biopsy samples and 17 age-matched controls (21). The discrepancies between the results of these valuable studies may be due to geographic differences, technical reasons, a heterogeneous disease process, or variation in the patient population. It should also be considered that PCR method is an extremely sensitive molecular technique used in identifying the viruses and may lead to erroneous diagnosis. Moreover, it should be remembered that most of the studied viruses are ubiquitous and most of the people are infected by them at some point during their lifetime (15).
Human herpesvirus type 8 (HHV-8), is the proposed etiological agent of Kaposi's sarcoma (22). Pleural effusion lymphoma and Castleman disease belong to the diseases associated with HHV-8 (23-24). Recently, Pulkkinen et al. detected Herpesvirus-specific DNA in the large majority of a series of IPF/UIP cases and other lung interstitial diseases (7). We have therefore considered analyzing the presence of this virus on our own IPF series as well by means of PCR, but we could not find a statistically significant association. While there is some evidence suggesting a link between HHV8 infection and IPF, proof of direct association is largely lacking. The limitation of this study was the sample size and the sensitivity of PCR method. Therefore, study of IPF series with a much larger sample size and with a more sensitive method could be more reassuring to conclude a result on this subject.
In conclusion, as we could not find EBV and HHV-8 DNA in our series of IPF patients, we suggest that EBV and HHV-8 may not be present in IPF samples. However, further studies with more samples and more sensitive methods are required to further elucidate this matter.
Mashhad University of Medical Sciences (MUMS) was the sponsor providing funding (T: 67470). This study was approved by the institutional review board (IRB) and ethics committee of MUMS.
The authors declared that there is no conflict of interest regarding the publication of this article.
| Article View | 1,594 |
| PDF Download | 1,066 |