Document Type : Original Research
Authors
1 Dept. of Clinical Sciences, Faculty of Veterinary Medicine, Shahid Bahonar Univercity of Kerman, Kerman, Iran
2 Dept. of Pathobiology, Faculty of Veterinary Medicine, Shahid Bahonar University of Kerman, Kerman, Iran
3 Veterinary Medicine, Faculty of Veterinary Medicine, Shahid Bahonar University of Kerman, Kerman, Iran
Keywords
Subjects
Introduction
Any restriction in blood flow to tissues can rapidly damage metabolically active tissues; however, there is evidence that restoration of blood supply to the tissue can cause additional damage known as ischemia-reperfusion injury (IRI) (1, 2). Renal I/R injury is considered as the main cause of acute renal failure in both native and grafted kidneys (3). A transient drop in total or regional blood flow to the kidney may result in ischemic acute renal failure syndrome. Both vascular and tubular factors are involved in the pathophysiology of I/R injury (1). Ischemia leads to capillary permeability, which is primarily mediated, by the action of reactive oxygen agents (ROS) and polymorponuclear neutrophils (4-7). Different strategies have been recently applied to reduce the ischemia and reperfusion injury to the graft (8). “Thus, ameliorating I/R-induced microcirculatory disturbance may be achieved by various means, including scavenging peroxides, inhibiting adhesion molecule expression in leukocyte, platelet and vascular endothelium, impeding adhesion of leukocyte and platelet to vascular endothelium, alleviating mast cell degranulation and depressing release of inflammatory mediators” (9).
Vitamin C (ascorbic acid) is a multifunctional nutrient involved in numerous biologic functions including collagen formation, biosynthesis of neurotransmitter, reductive production of vitamin E, and copper and iron reduction. Scientists have taken an interest in vitamin C since it is demonstrated as an antioxidant neutralizing free radicals (10). By rapid electron transfer, vitamin C acts directly as a water-soluble endogenous antioxidant, which scavenges aqueous reactive oxygen species (11).
Vitamin E is a lipid-soluble micronutrient and is mostly associated with plasma lipoproteins. It is a potent antioxidant acting as s peroxyl radical scavenger via breaking the chain reaction of lipid peroxidation (12). Vitamin E, in addition to inhibition of oxidative modification of low-density lipoprotein, may protect vascular walls from deposition of atherogenic oxidized low-density lipoprotein and proliferation of smooth muscle cells. Vitamin E as well as any other free radical scavenger may act as a second line of defense (12).
Corticosteroids like Hydrocortisone are often considered as the gold standard anti-inflammatory drugs.
Corticosteroids may protect against I/R injury by reduced complement activation, diminished pro-inflammatory (TNF-a, IL-1b, IL-6, and IL-8) and increased anti-inflammatory (IL-10) cytokine production, attenuated leukocyte activation and adhesion molecule expression, and sequestration (13).
This study has tried to use vitamin C, vitamin E, hydrocortisone and their combination for prevent the adverse effects of free radicals released from ischemic tissue after reperfusion in renal tissue.
Materials and Methods
Animals and experimental groups
The experimental protocols were approved by the Research Ethic Committee of the Shahid Bahonar University of Kerman, Iran .
Overall, 30 wistar strain male rats, weighing 280-300 gr were selected for the study. They were housed in plastic cage with filter tops under controlled conditions of 12-h light/12-h dark cycles 50% humidity at 28 °C. Animal were allowed a standard feed and water ad libitum. They were acclimatized to the environment for 1 week prior to experimental use. The animals were randomly divided into 6 groups (n=5 rats in each group) include: Sham, I/R injury, Vit C, Vit E, hydrocortisone, and Combination of Vit C & E and hydrocortisone.
To induce ischemia, after induction of anesthesia with Ketamine (90 mg/kg, IM) and Xylazine (90 mg/kg, IM), each rat was placed in dorsal recumbent position. After routine preparation for surgical site, a laparotomy was performed using a midline skin incision. The bowels were partly exteriorized outside the peritoneal cavity to expose the abdominal aorta. Renal ischemia was induced by clamping the abdominal aorta with a vascular clamp above the renal artery. The abdomen was temporarily closed and after 45 min, the clamp was removed to reperfuse the kidneys. One hour after reperfusion, the animals were sacrificed with intracardiac injection of sodium thiopental, then the abdomen was reopened and the left kidney was removed. The animals in-group Vit C, received vitamin C (Daroupakhsh Co., Tehran, Iran) at dose of 50 mg/kg intravenously, at the same time with removing the clamp. The animals in group Vit E, received vitamin E (Osvah Co., Tehran, Iran) at dose of 20 mg/kg intramuscularly, 15 min before removing the clamp. The animals in group Hydrocortisone, received an intravenous injection of hydrocortisone (Rotex-medica, Trittau, Germany) at dose of 50 mg/kg intravenously, at the same time with removing the clamp. The animals in group Combination, received intravenous injection of vitamin C and hydrocortisone, and intramuscular injection of vitamin E at the mentioned time point and dose. The rats in group I/R underwent the same surgical procedure and did not receive any treatment. In the group sham, the abdominal cavity was opened; the kidneys and aorta were manipulated without applying the clamp, and the animals did not receive any treatment. Both intramuscular and intravenous injections of medications or normal saline (at the same volume) were carried out in each animal of all groups.
The left kidneys were collected at 60 minute after reperfusion time and processed for microscopic study. The tissues were fixed by dipping in 10% phosphate-buffered formalin, afterwards the samples were embedded in paraffin wax. Five-micrometer sections of the samples were cut and stained with hematoxyline and eosin, and then examined in a blinded fashion under light microscope.
Tubular degeneration, necrosis, tubule interstitial nephritis, and total histological scores were evaluated semi-quantitatively (14) (Table 1)
Tubular degeneration (TD): in the cytoplasm of the proximal tubule epithelial cells, stained bodies of various sizes and vacuolization containing acidophilus were considered as TD.
Tubular necrosis (TN) defined as loss of epithelial cells of the nucleus, dark acidophilic cytoplasm, loss of tubular epithelial cells into tubular lumen, and acellular sections of tubules.
Tubulointerstitial inflammation (TIN) defined as infiltration of inflammatory cells in perivascular and interstitial areas.
Total histologic score (THS): TD/2 + TN + TIN/2, respectively.
Table 1: Definition of scores for tubular degeneration (TD), necrosis (TN), tubule interstitial nephritis (TIN), and total histological scores (THS)
|
TD & TN |
|
|
Score 0 Score 1 Score 2 Score 3 Score 4 |
Absence of TD/TN Mild: small and a few focus TD/TN in immediately beneath the capsule (0%–10) Moderate: for a few focal focus TD/TN and along the tubular segment (10%–25) Severe: diffuse and significant TD/TN along the tubular segment (% 25–50) Very severe: TD/TN was greater than 50% |
|
TIN |
|
|
Score 0 Score 1 Score 2 Score 3 Score 4 |
Absence of TIN Mild TIN: a few pieces of infiltration concentrated on perivascular area (0–5%) Moderate TIN: infiltrations involved in cortical interstitial and many focal areas (5–10%) Severe TIN: diffuse and significant infiltration areas (15–25%) Very severe TIN: TIN was greater than 50% |
|
THS |
|
|
Score 0-2 Score 2-5 Score 5-8 Score >8 |
Normal THS Mild THS Moderate THS Severe THS |
Statistical analysis
Obtained data were expressed as median and range for all groups. Scored values were analyzed using nonparametric Mann–Whitney U test. The SPSS software version 16 (SPSS Inc., Chicago, IL, USA) was used in order to perform analyses. P<0.05 was considered significant.
Results
The kidney sections from the group sham revealed the normal preserved kidney structure and showed normal tubular epithelial cells and glomeruli without any signs of degeneration and necrosis. All histopathologic scores in this group was considered zero.
Tubular damages related to ischemia/reperfusion were extensive in I/R group. Proximal and distal tubules showed severe vacuolar degeneration and necrosis. Degenerative changes demonstrated with cell swelling due to formation of cytoplasmic vacuoles. Epithelial cells detached from the basement membrane and loss their brush border in proximal tubules. Pyknotic and disappearing of nucleus was observed in the necrotic tubules. Glomeruli were atrophic and had dilated urinary space (Fig.1). A marked congestion and hemorrhage occurred in this group.
In contrast, renal sections obtained from animals treated with vitamin C exhibited a noticeable reduction in the severity of tubular injury. Group Vit C had minimal histopathologic alterations and only mild degenerative changes occurred in the epithelium of proximal tubules (Fig. 2). Tubular injury in both vitamin E and hydrocortisone groups showed decrease renal damages in comparing with group I/R (Fig. 3, 4). The protective effect of combination group was more than other experimental groups. The kidney showed histological structure similar to normal and sham groups. No signs of cell swelling and necrosis were observable (Fig. 5).

Fig. 1 Group I/R. Severe vacuolar degeneration and necrosis (Hematoxylin and Eosin Staining ×400)

Fig. 2 Group Vit C. Mild degenerative changes in the epithelium of proximal tubules (Hematoxylin and Eosin Staining ×400)
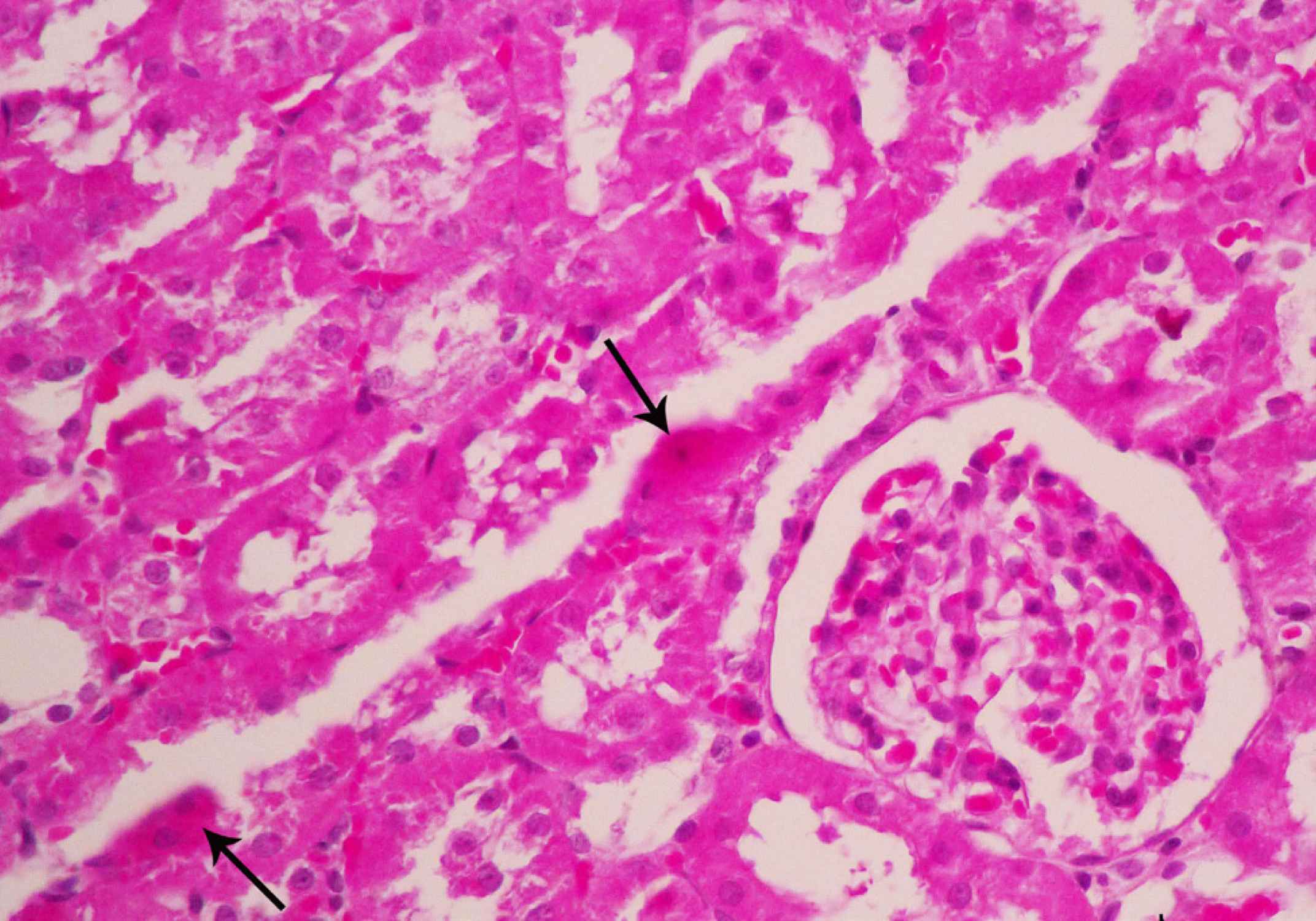
Fig. 3 Group Vit E. The kidney section shows tubular degeneration and scare cellular necrosis (arrows) (Hematoxylin and Eosin Staining ×400)

Fig. 4 Group Hydrocortisone. Moderate vacuolar degeneration of tubular epithelium (Hematoxylin and Eosin Staining ×400)

Fig. 5 Group Combination. The kidney shows preserved normal histologic structure (Hematoxylin and Eosin Staining ×400)
The histological scores (median and range) of tubular degeneration 3[3-4], tubular necrosis 3[2-3], tubulointerstitial inflammation 1[0-1] and total histological score 5[4-5.5] in group I/R significantly increased compared to the group sham (P<0.05).
Score of tubular degeneration in groups Vit C 1 [1-2], Vit E 2[1-2], Hydrocortisone 1[1-3] and Combination 1[0-1] significantly decreased compared to the group I/R; and there were significant differences between them and sham groups.
The given scores for tubular necrosis showed that there was significant differences in groups Vit C 0[0-0], Vit E 1[0-1], Hydrocortisone 0[0-1] and Combination 0[0-0] compared to the group I/R. In addition, significant differences only observed in group Vit E in comparison with group sham.
There was no significant difference in tubulointerstitial inflammation score between all treatment groups and groups sham and I/R (P>0.05).
Analysis of total histological scores showed this parameter significantly decreased in groups Vit C 0.5[0.5-1], Vit E 1.5[1-2], Hydrocortisone 0.5[0.5-3] and Combination 0.5[0-1] compared to the group I/R; and there were significant differences between them and group sham.
Comparison of the recorded scores between the treatment groups showed score of tubular degeneration in group Vit E was significantly more than group combination, and tubular necrosis and total histologic scores in group Vit E were significantly more than groups Vit C and combination.
Groups Combination and Vit C showed more effective and group Vit E showed less effective in protecting of renal tissue against the renal I/R injuries.
Table 2: Histopathological evaluation of tubular degeneration (TD), necrosis (TN), tubule interstitial nephritis (TIN), and total histological scores (THS) in different groups
|
Groups |
Histopathologic scores (median & range) |
|||
|
TD |
TN |
TIN |
THS |
|
|
Sham |
0(0) |
0(0) |
0(0) |
0(0) |
|
I/R |
3(3-4)a |
3(2-3)a |
1(0-1)a |
5(4-5.5)a |
|
Vit C |
1(1-2)ab |
0(0)b♣ |
0(0-1) |
0.5(0.5-1)ab♣ |
|
Vit E |
2(1-2)ab٭ |
1(0-1)ba٭♣ |
0(0-1) |
1.5(1-2)ab٭♣ |
|
Hydrocortisone |
1(1-3)ab |
0(0-1)b |
0(0-1) |
0.5(0.5-3)ab |
|
Combination |
1(0-1)ab٭ |
0(0)b٭ |
0(0-1) |
0.5(0-1)ab٭ |
- (a) and (b) letters show significant difference (P< 0.05) in comparison with sham and I/R groups, respectively.
- Same symbols show significant difference (P< 0.05) between treatment groups.
Discussion
I/R injury result in damage to endothelial and parenchymal cells, both reactions of granulocytes and macrophages, including complement, coagulation factors, free radicals, and nitric oxide. The most important free radical in aerobic environment is ROS. These include superoxide anion O2-, singlet oxygen O-, hydrogen peroxide H2O2, nitric oxide NO-, its conjugate acid HOO-, lipid and proteins, carbohydrates and nucleic acids In addition, I/R injury, by activating macrophages, plays a role in the release of cytokines, which, in turn, affects leukocyte activation, transmigration, and target cell adhesion. The large amounts of leukocytes entering the extravascular space immediately after reperfusion produce enormous quantities of ROS (5, 15).
A rapid and extensive oxidative stress is resulted from renal I/R damage and subsequent acute renal failure. The secondary destruction following renal injury will be minimized by decreasing the degree of oxidative stress (16, 17). The cellular defensive response to overproduction of ROS following renal I/R indicates the increased antioxidant capacity and treatment with antioxidant agents (18).
Ascorbic acid is powerful protective antioxidants, which neutralize the deleterious effects of antioxidants like H2O2 on endothelium, and prevents microvascular dysfunction (19-21). Vitamin C by inhibit reactive oxygen species via rapid aqueous-phase electron transfer, thereby reducing adhesion of neutrophils to endothelium so it decreases both the generation of oxygen free radicals by neutrophils and subsequent lipid peroxidation. When the neutrophils are chemo attracted to the site of I/R injury, vitamin C pretreatment may decrease subsequent tissue damage (22). Vitamin C can effectively improve cardiovascular function after coronary artery bypass grafting surgery (23). Ascorbic acid has been used in rabbits to protect against corneal damage induced by free radicals. In addition, it has also been administered in pigs to improve the renal hemodynamics as well as reduce oxidative stress, inflammation and fibrosis in the ischemic kidney. Ascorbic acid is a water soluble and easily administered antioxidant that is available at a low price (24). The protective effects against increase of lipid peroxidation resulted from I/R were shown in rats treated with different dosages (range, 30-100 mg/kg) of vitamin C (25). As mentioned by Niki et al. (26), vitamin C has antioxidant effect of the superoxide and hydrophilic radicals. It also plays an important role in limiting lipid peroxidation and scavenges reactive oxidants produced immediately after reperfusion (27). The histopathology finding in the current study has showed that intravenous injection of single dose of vitamin C (50 mg/kg) immediately after reperfusion of blood flow noticeably reduce I/R injuries in renal tissue.
Corticosteroids are routinely and effectively used in treatment f various inflammatory and autoimmune disorders. Corticosteroids are synthetic analogues of the steroid hormones that are naturally produced in the adrenal cortex. Mineralocorticoids are primarily involved in electrolyte and water balance regulation by affecting ion transport in epithelia cells of the renal tubule. Glucocorticoids, on the other hand, are mainly involved in metabolism of carbohydrate, fat and protein, and have anti-inflammatory, anti-proliferative, immunosuppressive and vasoconstritive effects (28). Administration of corticosteroids is associated with improved cardiovascular performance and reduced perioperative morbidity in precardiopulmonary bypass (29), and reported that pretreatment with steroids was protective against spinal cord ischemia in a canine model (30, 31), but the risk of increased infection and poor wound healing, besides the controversy regarding efficacy and dosing regimens, have limited the routine use of corticosteroids (32). The results of microscopic study in the present study showed that intravenous administration of hydrocortisone at the time of reperfusion significantly decreased the adverse effects of I/R injuries on the renal tissue.
Vitamin E and its analogs have been the subject of many studies and are shown to have potent oxygen scavenging properties as well as membrane peroxidation inhibitory activity, continued to be studied more for their role in the management of I/R injury (33). NF-κB is activated during the ischemic and subsequent reperfusion events (34). NF-κB is activate by the cellular cascade occurred during reperfusion, which is initiated by cytokine and ROS-dependent mechanism. In recent studies, vitamin E has been shown to inhibit the transcriptional activity of NF-κB and block the binding activity of NF-κB at the nuclear level. Vitamin E exerts its effects as an antioxidant as well as other biological effects (35); moreover, it is shown to decrease the occurrence of ventricular fibrillation during reperfusion (36). Improved recovery of cardiac function following I/R, as measured by heart rate and left ventricular pressure, was also reported in rats when pretreated with vitamin E (37). Muscle viability and blood flow 24 hours following reperfusion can be increased when treated with vitamin E (38). Vitamin C is not capable of scavenging the peroxyl radical in the lipid phase; however, it is effective in regenerating vitamin E from the tocopheroxyl radical, and therefor necessary in recycling of vitamin E (39). According to the histopathologic evaluation, treatment with vitamin E has not considerably amelioration effects against tissue injury following ischemia and reperfusion event in the kidney, in comparison with other medications used in the current study. In a study by Gunel, ascorbic acid showed more antioxidant effect on intestinal ischemia reperfusion injury in rabbit, compared with vitamin E (39). In the current study, single dose of Vit E (20 mg/kg) was administered intramuscularly fifteen minutes before reperfusion of blood flow to the kidney. The difference in effectiveness of vitamin E may be because of the different application manner in pretreatment and reperfusion therapy and because of the intramuscular administration of this drug.
Conclusion
The results showed the combination of Vit C and E, and hydrocortisone may be sufficient to more efficiently prevent subsequent oxidative stress in renal I/R injury and improve their function after I/R. Smultaneous administration of this combination could show a synergy against deleterious effects of I/R injuries in kidney.
Acknowledgements
This work was supported financially by a Grant for Scientific Research from Vice Chancellor of Research of Shahid Bahonar University of Kerman, Iran.
Conflict of interest
The authors declare that there is no conflict of interests.
| Article View | 3,161 |
| PDF Download | 1,727 |